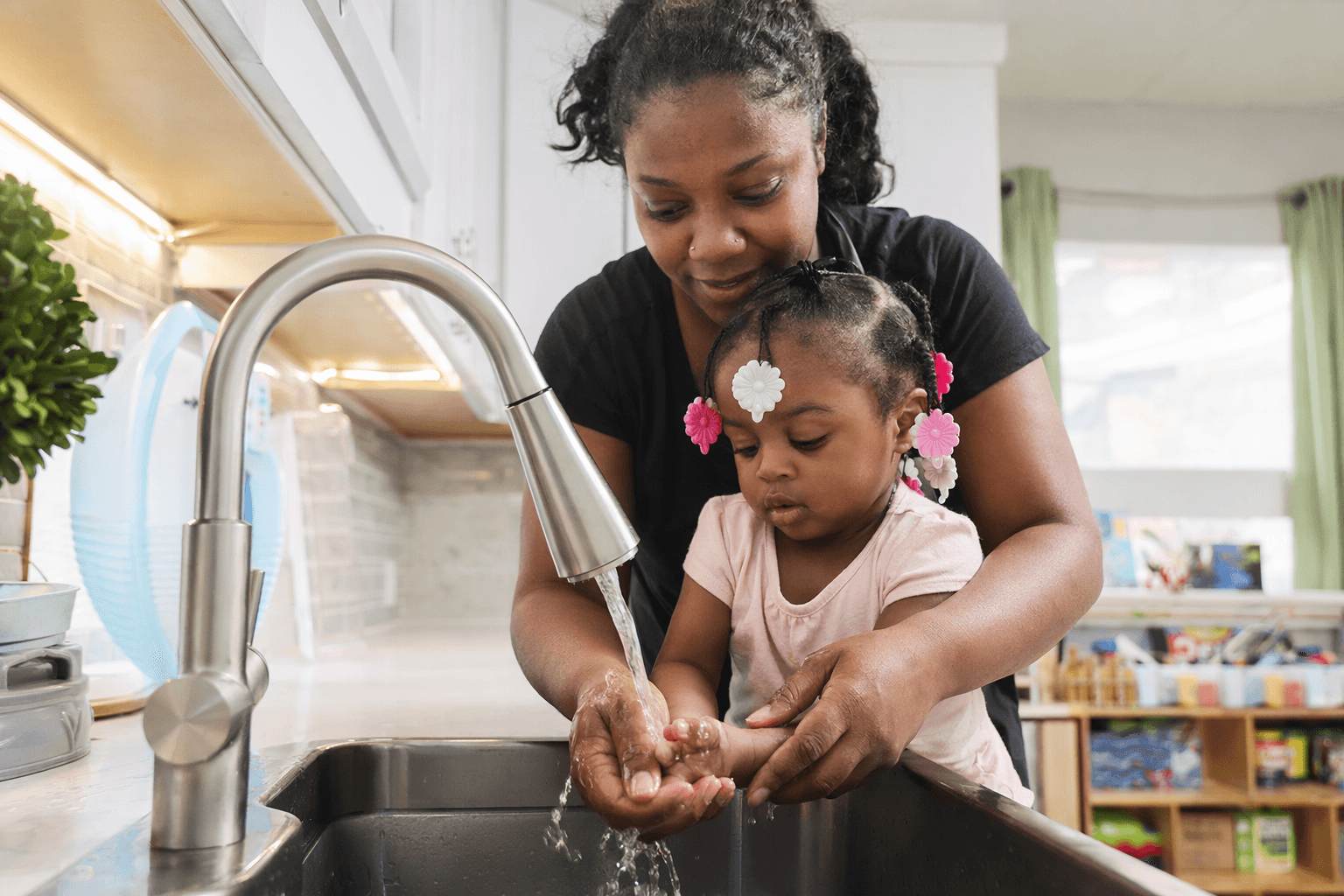Elbridge siblings hear for first time after FDA-approved gene therapy
Raegan and Emrys McLaughlin-Eller heard their mother’s voice after gene therapy, turning a rare Elbridge diagnosis into a medical first.

Raegan and Emrys McLaughlin-Eller heard what their family had waited years to hear: their mother’s voice. For Erin McLaughlin-Eller and Joshua McLaughlin-Eller of Elbridge, that moment marked a life-changing shift from silence to sound for their 5-year-old daughter and 3-year-old son.
Both children were born with Auditory Neuropathy Spectrum Disorder, or ANSD, a rare condition that CNY Central reported affects only about 20 to 50 children in the United States each year. Erin first noticed Raegan’s hearing delay around the summer of 2022, and Emrys was born in August 2022. The family enrolled Raegan in a hearing study before the gene-therapy treatment, after testing at home suggested neither child was responding to their names or loud sounds.
The children underwent surgery in January 2025. WSYR-TV reported that Emrys began hearing within weeks, around mid-February 2025, and Raegan began hearing by mid-April 2025. The practical changes were immediate and ordinary in the best possible way: Erin said the children could hear horns, sirens, cars coming and their names, not just speech. They could also hear their mother say, "I love you."
Their progress came as the U.S. Food and Drug Administration approved Otarmeni, also known as lunsotogene parvec-cwha, on April 23, 2026. The FDA said it was the first-ever dual adeno-associated virus vector-based gene therapy and the first gene therapy approved under the Commissioner’s National Priority Voucher pilot program. The agency said the approval came 61 days after the biologics license application was filed, tying it for the fastest BLA approval in modern FDA history.

The therapy is indicated for patients with severe-to-profound and profound sensorineural hearing loss linked to biallelic OTOF variants. The FDA said OTOF variants account for about 2% to 8% of inherited, non-syndromic hearing-loss cases, and that genetic mutations cause about half of congenital hearing loss. The American Society of Gene & Cell Therapy said this is the first approved gene therapy to directly target a genetic form of hearing loss, a milestone that could matter far beyond one household in Onondaga County.
Regeneron Pharmaceuticals developed the treatment and said it would offer Otarmeni free to U.S. patients. In the CHORD trial, 20 participants received injections and 16 met the primary endpoint of hearing improvement. For the McLaughlin-Ellers in Elbridge, though, the breakthrough was simpler and more personal: their children heard them, and the world changed.
Know something we missed? Have a correction or additional information?
Submit a Tip