Boy nearly dies after vitamin D overdose from faulty drops batch
Seven-year-old Roo was given vitamin D drops from a batch that was about seven times too strong, leaving him with acute kidney injury and a near-fatal overdose.
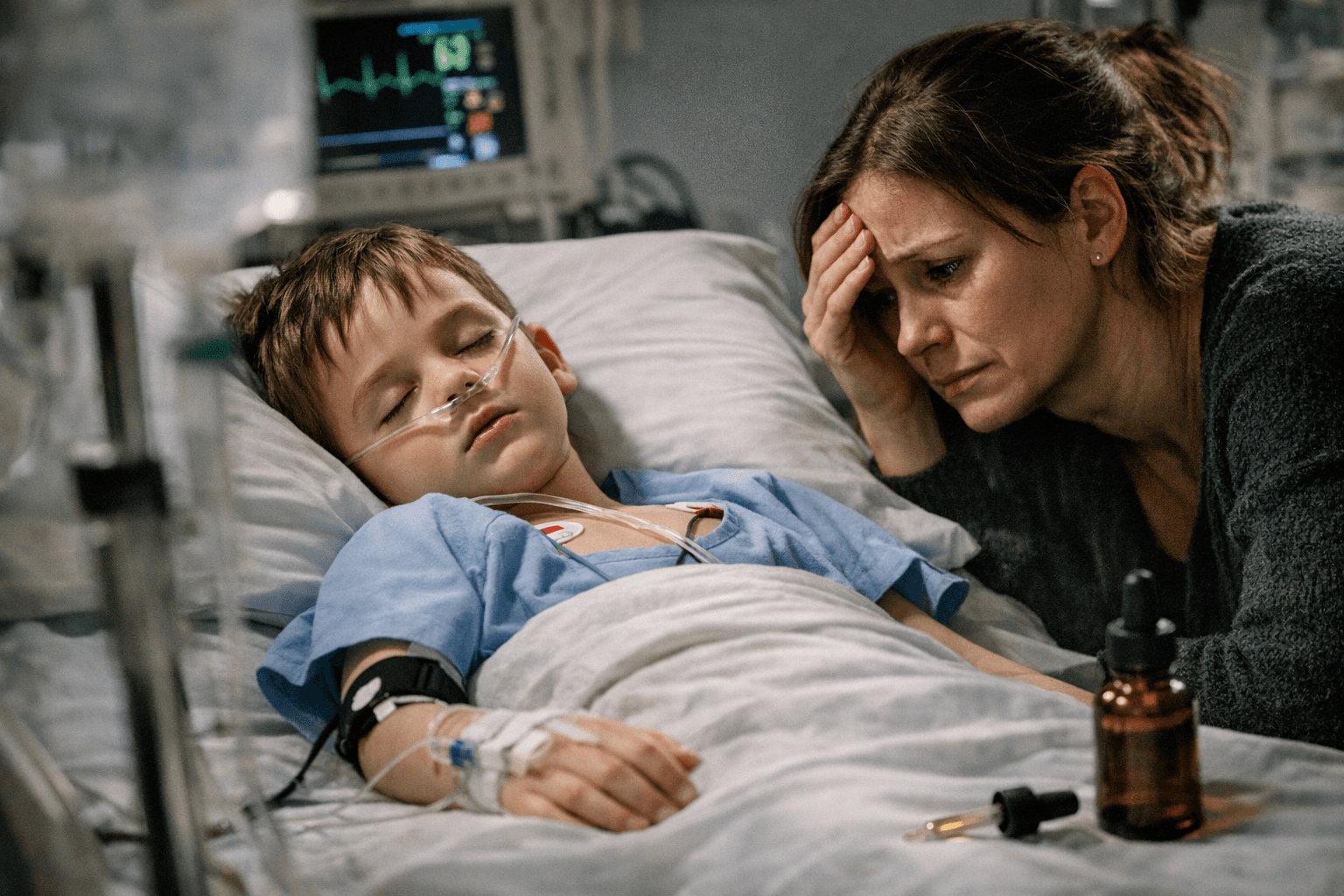
Seven-year-old Roo was left with acute kidney injury after vitamin D3 drops prescribed for his leg pain turned out to be about seven times stronger than they should have been. The bottle came from one of two bad batches distributed across the United Kingdom, turning a routine supplement into a life-threatening dosing failure and raising sharp questions about labeling, oversight and follow-up.
Roo had been prescribed a 12-week high-dose course of vitamin D3 drops in December 2024 to ease intense pain in his legs. Before the cause was identified, he lost weight and drank excessively, symptoms that led doctors and his parents to fear he might have a brain tumour. His mother, Alaina, said more than a year later that he had still not fully recovered.
Vitamin D is essential for healthy bones, teeth and muscles because it helps regulate calcium and phosphate, but higher-dose vitamin D products prescribed by doctors are still classed as food supplements rather than medicines in the United Kingdom. That means they are monitored by the Food Standards Agency rather than the Medicines and Healthcare products Regulatory Agency, a split in responsibility that sits at the center of this case.
On 8 January 2025, the Food Standards Agency recalled TriOn Pharma’s Aactive D3 Drops and Aactive D3 Solution after finding they contained higher levels of vitamin D3 than were written on the label. The recall was updated the following day with product images. The concern was not only that the dose was wrong, but that a supplement prescribed to a child for a minor complaint could pass through ordinary prescribing and dispensing checks while carrying a hidden toxic load.
The danger extended beyond Roo. Another child, Kayan Khan, had been prescribed the same brand a month before Roo began his course and was admitted to hospital in February 2025 with high calcium levels and reduced kidney function. A letter from Sheffield Children’s NHS Foundation Trust said doctors struggled to control his calcium levels and found vitamin D intoxication far above what would be expected from standard supplementation.
A leading expert said Roo would have died if he had completed the prescribed course. The case now stands as a warning about every step in the chain, from prescribing and labeling to batch testing and clinical follow-up, because the failure of one weak link was enough to poison a child.
Know something we missed? Have a correction or additional information?
Submit a Tip

