Eli Lilly to buy Kelonia Therapeutics for up to $7 billion
Lilly is paying up to $7 billion for Kelonia to chase in vivo CAR-T, a one-shot therapy that could cut out apheresis, factory steps and chemotherapy.

Eli Lilly is betting that the next big leap in cancer cell therapy will happen inside the patient’s body, not in a manufacturing plant. The company said it agreed to acquire Boston-based Kelonia Therapeutics for up to $7.00 billion in cash, including a $3.25 billion upfront payment, giving Lilly a deeper foothold in genetic medicine and next-generation cancer research.
The prize is Kelonia’s lead program, KLN-1010, a Phase 1 therapy for relapsed or refractory multiple myeloma that targets BCMA, a marker expressed on myeloma cells. Kelonia describes the treatment as a one-time intravenous in vivo CAR-T therapy built on its proprietary gene placement system, or iGPS, which uses engineered lentiviral-based particles to reach T-cells inside the body and generate CAR-T cells without the usual ex vivo manufacturing process.
That distinction matters for both patients and drugmakers. Conventional CAR-T therapies can produce striking results, but they are complicated to make, slow to deliver and expensive to scale. Kelonia says KLN-1010 is administered without apheresis, ex vivo manufacturing or lymphodepletion chemotherapy, removing three of the biggest barriers in today’s CAR-T workflow. Independent reviews of in vivo CAR-T have argued that the approach could reduce the complexity, timeline and cost burden that have kept cell therapy largely confined to specialized centers and narrow indications.
Lilly said the acquisition is intended to expand its genetic medicine capabilities and broaden its work in in vivo gene delivery and integration technology. The move also reflects a wider industry push to turn CAR-T into a more scalable platform, rather than a bespoke treatment available only to a small slice of patients. Lilly oncology chief Jacob Van Naarden said autologous CAR-T therapies have improved outcomes, but manufacturing, safety and access barriers mean only a fraction of eligible patients receive them.
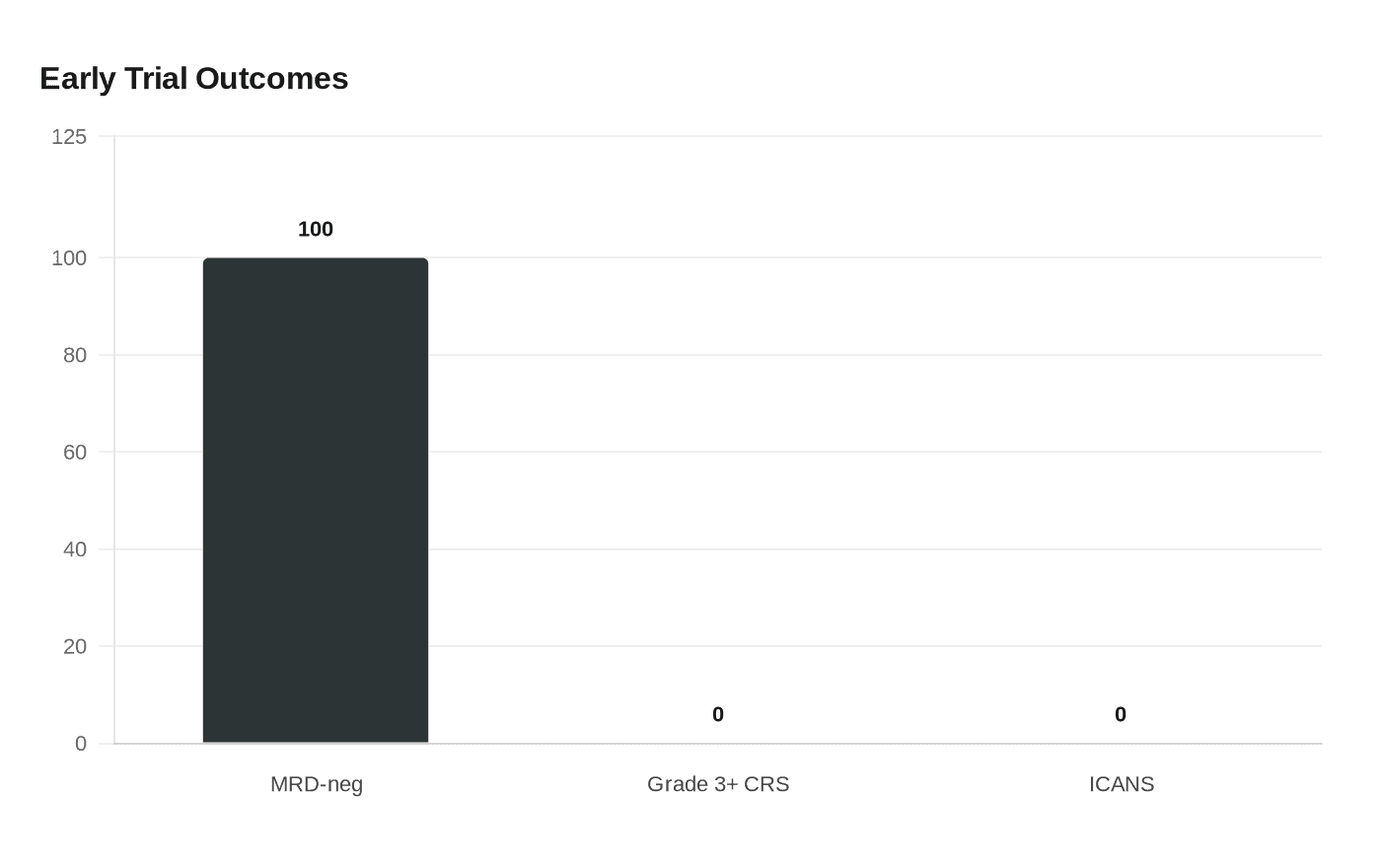
The early clinical signals are encouraging, but they are still early. Kelonia said data presented at the American Society of Hematology meeting in 2025 showed a 100% MRD-negative response rate in the first four patients, with responses maintained through the longest follow-up of five months. The company also reported no grade 3 or higher cytokine release syndrome and no ICANS. Lilly said the data are promising and suggested the platform could have broader uses across diseases where in vivo cell therapy might help.
For investors, the real question is whether Lilly has found a transformative platform or paid a premium for a technology that is still proving itself. A $7 billion price tag implies confidence that in vivo CAR-T can solve one of biotech’s hardest operational problems: how to turn a powerful personalized therapy into something faster, simpler and more widely available. The answer will depend on whether Kelonia can turn a small Phase 1 signal into durable responses, safer delivery and a manufacturing model that can change the economics of cell therapy.
Know something we missed? Have a correction or additional information?
Submit a Tip

