FDA Clears Waters At-Home HPV Kit, Expanding Cervical Cancer Screening Access
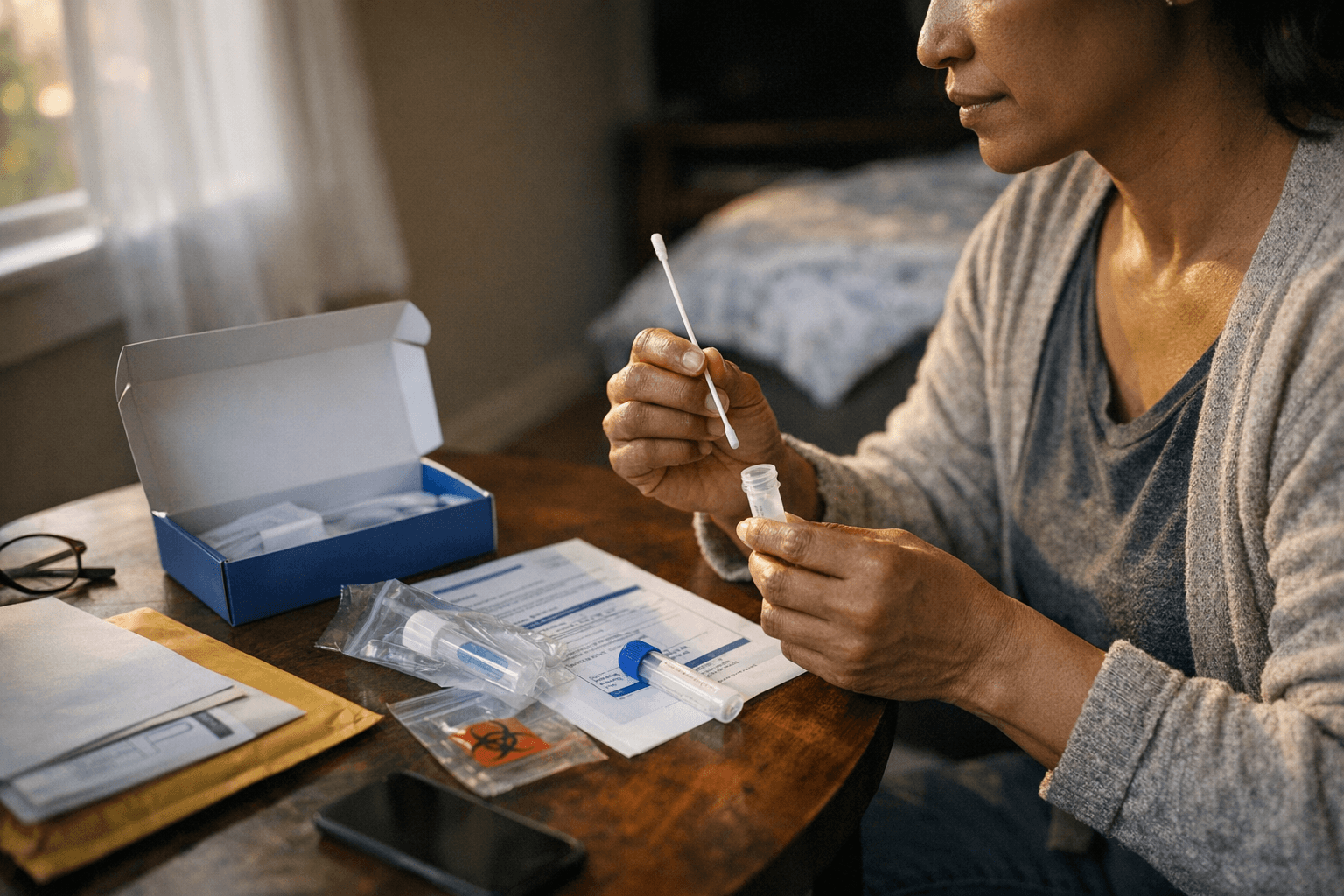
Waters Corp.'s FDA-cleared Onclarity HPV kit lets patients collect cervical cancer screening samples at home and mail them to labs, targeting chronically under-screened populations.
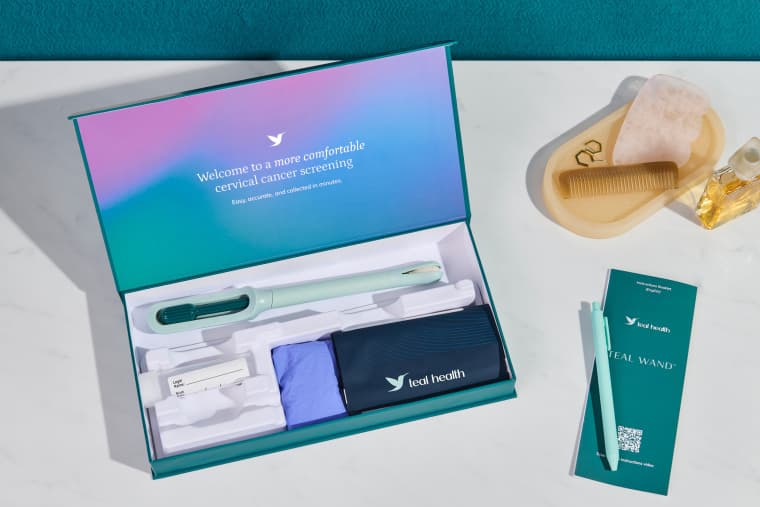
Waters Corporation secured FDA clearance Wednesday for its Onclarity HPV Self-Collection Kit, enabling people with a cervix to collect samples at home and mail them to certified laboratories for high-risk HPV testing, in what the Milford, Massachusetts company called the most comprehensive at-home cervical cancer screening tool currently cleared in the United States.
The clearance pairs the Onclarity HPV Self-Collection Kit with the BD Onclarity HPV Assay, manufactured by Becton, Dickinson and Company. Together, the two products are designed to integrate with automated BD COR laboratory workflows, allowing central labs to process self-collected, mailed samples at high throughput without manual handling at each step. Waters said it plans to establish partnerships with laboratories, health systems, and providers to expand nationwide distribution and testing capacity.
The regulatory decision targets a persistent gap in cervical cancer prevention. Clinic-based Pap smears and HPV tests have long been the standard pathway, but under-screened and never-screened populations account for a disproportionately large share of cervical cancer diagnoses and deaths. Waters framed the clearance as a direct response to that inequity, arguing that home collection can remove barriers including lack of transportation, inconvenient clinic hours, embarrassment, and distrust of health systems that keep many patients from presenting for routine screening.
A positive result from the at-home kit still requires clinical attention. Patients who test positive for high-risk HPV strains need further evaluation, which can include colposcopy or additional clinical procedures. Clinicians have consistently emphasized that any expanded screening program must include clear pathways for result navigation and counseling alongside sample collection, because detection without follow-through delivers incomplete protection.
Waters' shares climbed in early trading after the announcement, reflecting investor confidence that FDA-cleared home diagnostics can expand addressable markets and drive recurring laboratory test volume. The commercial logic embedded in the announcement centered on high-throughput scalability: by funneling self-collected samples into automated workflows, laboratories and health systems can absorb substantially larger testing volumes without proportional increases in staffing or overhead.
For policymakers, the clearance sharpens questions that home diagnostic expansion consistently raises: what reimbursement structures will govern kit distribution and lab processing, how labs will manage surging sample volume, and what mechanisms will ensure patients who test positive receive timely care. Those logistical and policy frameworks will ultimately determine whether broader access to screening translates into measurable reductions in invasive cervical cancer incidence, particularly in rural, low-income, and underserved communities where the barriers Waters cited have historically been the steepest and the screening gaps the widest.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip