Genentech says fenebrutinib cut relapses 51% in pivotal RMS trial, signals regulatory filing
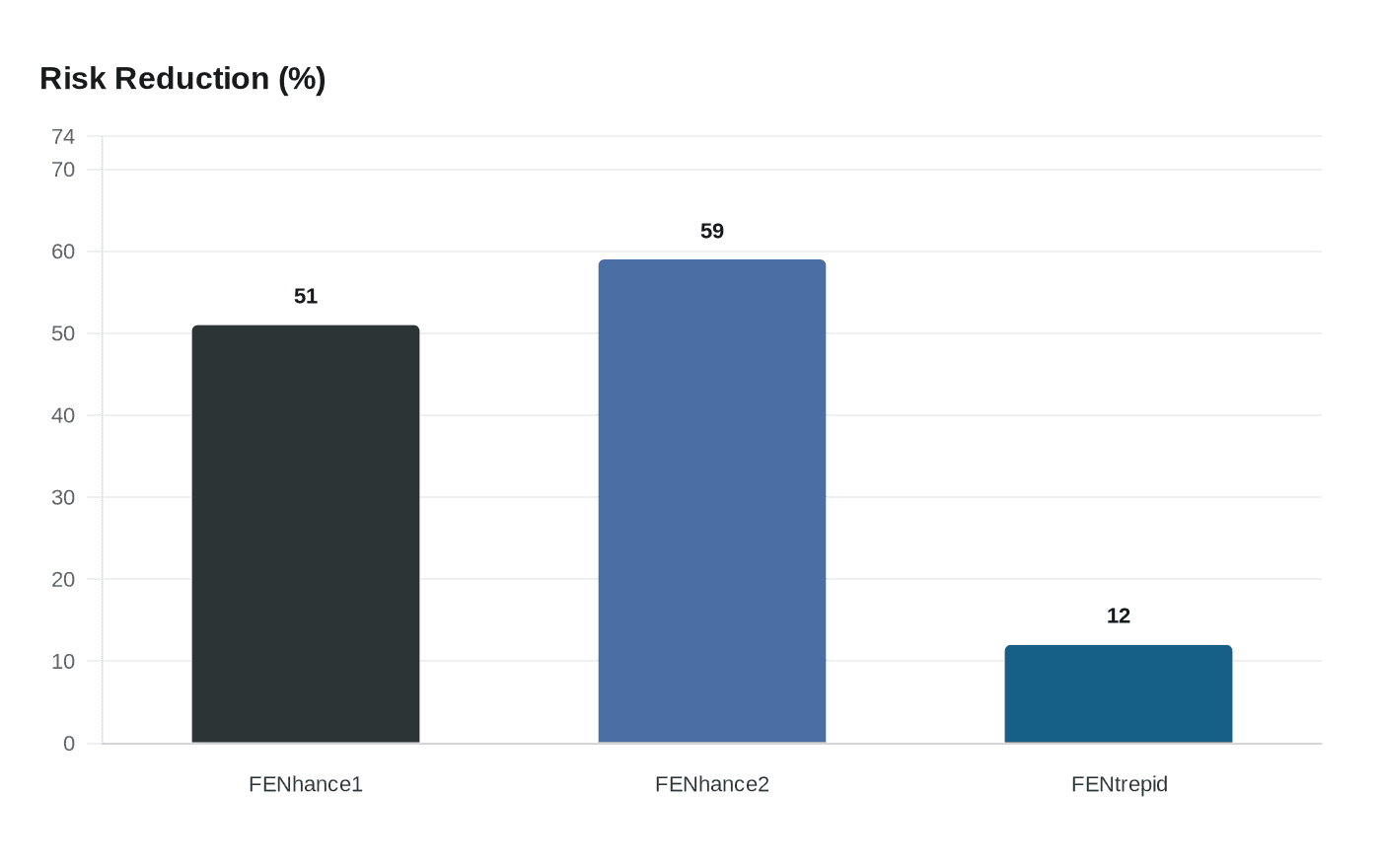
Genentech and Roche said FENhance 1 met its primary endpoint with a 51% ARR reduction versus teriflunomide and that the company will compile all Phase III data for regulatory submission.

Genentech said on March 1, 2026 that FENhance 1, the pivotal Phase III study of investigational BTK inhibitor fenebrutinib in relapsing multiple sclerosis, met its primary endpoint, reporting a 51% reduction in annualized relapse rate versus teriflunomide over at least 96 weeks of treatment. Roche issued a Basel press release dated March 2, 2026 that described the result as clinically meaningful and statistically significant and said the company will compile the three Phase III datasets for regulatory submission.
The FENhance 1 readout completes a program that already included FENhance 2 and FENtrepid. Genentech and Roche had earlier reported that FENhance 2 showed a 59% ARR reduction versus teriflunomide. Roche and Genentech frame the combined RMS results as so large that, together, they “equate to approximately one relapse every 17 years,” a company-calculated comparison intended to convey the magnitude of effect.
The third trial, FENtrepid, tested fenebrutinib in primary progressive MS against Ocrevus (ocrelizumab). That study was reported as meeting its primary endpoint of non-inferiority; fenebrutinib reduced the risk of disability progression by 12% versus Ocrevus (hazard ratio 0.88; 95% confidence interval 0.75 to 1.03), with Kaplan-Meier curves separating as early as 24 weeks. Those data were presented as a late-breaking oral session at the ACTRIMS Forum 2026. Levi Garraway, Roche chief medical officer and head of global product development, said in earlier reporting that “Fenebrutinib represents the first potential scientific breakthrough for the PPMS community in over a decade,” and the company said it looks forward to advancing regulatory filings after FENhance 1 readout.

Safety and prior regulatory history remain central to assessing the program. Fenebrutinib was placed on partial FDA hold in 2023 after two cases of elevated liver enzymes in an MS trial, one of which met Hy’s Law criteria. Company statements and industry summaries say liver monitoring was implemented and no further Hy’s Law cases were reported thereafter. In FENtrepid, the safety profile was described as generally comparable to Ocrevus, with similar rates of infections and nausea; transient and reversible liver enzyme elevations were reported more frequently in the fenebrutinib arm but no cases meeting criteria for severe drug-induced liver injury were observed.
If regulators accept the combined Phase III package, fenebrutinib could alter clinical practice by offering an oral, brain-penetrant BTK inhibitor positioned by the company as a high-efficacy option for both relapsing and progressive disease. Ocrevus remains the only approved medicine for PPMS, and an oral alternative that shows benefit on progression endpoints could shift prescribing patterns and payer negotiations, particularly for patients seeking noninfusion options.
Roche said more than 2,700 patients and healthy volunteers have been treated with fenebrutinib across Phase I through III programs. The company reiterated intent to submit the totality of Phase III data to regulatory authorities but did not provide a specific filing date; prior reporting had suggested a filing could occur later in 2026 pending FENhance 1. Full trial-level details, including patient counts, absolute event rates, p-values and comprehensive safety tables, were not included in the topline announcements and will be crucial for regulators and clinicians to evaluate the drug’s benefit-risk profile.
Know something we missed? Have a correction or additional information?
Submit a Tip

