Hormone Patch Shortages Grow as Demand Surges, Mostly for Good Reasons
Estrogen patch prescriptions rose 86% since 2021, and the FDA's removal of a two-decade-old safety warning has pushed already-strained supplies into a nationwide crunch.
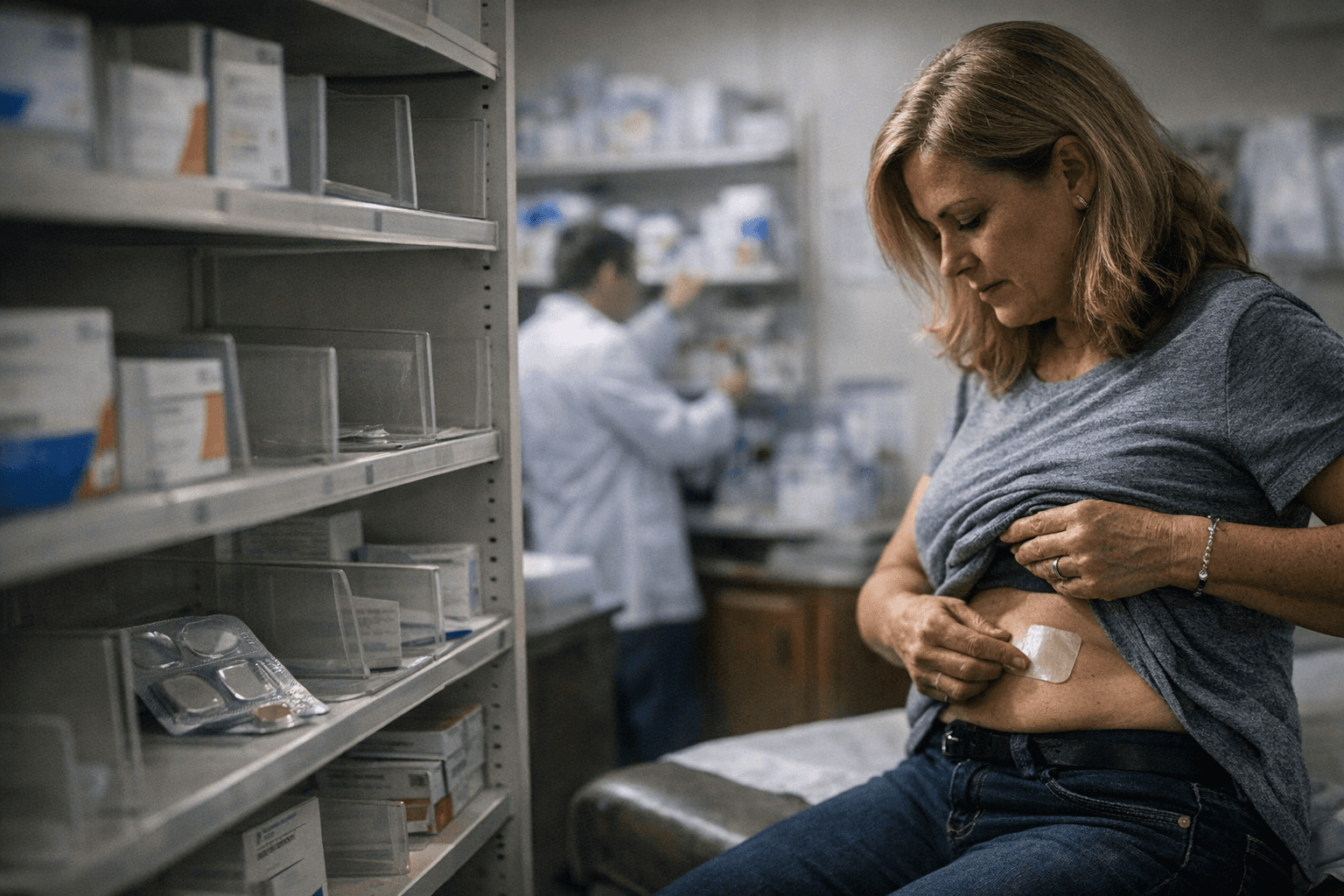
Women who rely on estradiol patches to manage menopause symptoms are calling pharmacies, driving between locations, and switching brands mid-treatment as a nationwide supply crunch drags into its second year. Transdermal estrogen patches, a commonly prescribed form of menopausal hormone therapy, are hard to find. For many patients, the disruption is not merely inconvenient: hot flashes, night sweats, and related symptoms return when doses are missed or treatments are abruptly switched.
Hormone replacement therapy prescriptions for women aged 50 to 65 increased 86 percent since 2021, according to Epic Research, which analyzes electronic health records nationwide. The sharpest acceleration came after November 2025, when the FDA removed the black box warning from all hormone therapy products containing estrogen. That warning, in place since 2003 following results from the large Women's Health Initiative study, had warned of increased risks of heart attacks, strokes, and pulmonary embolism. For years, it overstated those risks and discouraged both patients and clinicians from even discussing estrogen. The decision to remove it was evidence-based and long overdue, according to medical organizations that had been calling for the change for years.
The supply side was simply not ready. "There's just more demand for these medications, and I think it's exponentially risen over the last three or four years," said Dr. Rajita Patil, director of UCLA Health's Comprehensive Menopause Program. "I don't think that the supply chain was ready for that." Only a handful of companies produce estradiol transdermal patches. Major manufacturers include Noven Pharmaceuticals, Sandoz, and Mylan, with a small group of generic makers including Amneal and Zydus. When Bayer discontinued its Climara patch in late 2023, the already thin manufacturing base shrank further. When production slows or a product is discontinued, there is very little redundancy to buffer the impact. A disruption at even one manufacturer can ripple nationally.
Amneal Pharmaceuticals confirmed to NPR that "following the FDA's removal of boxed warnings on hormone replacement therapy, we have seen a significant increase in demand." The company added that the patches require specialized manufacturing and that there is "finite industry capacity" for producing them. Sandoz, the Switzerland-based maker of a widely used generic patch, said supply challenges were driven by a global shortage and "unprecedented demand that cannot be fully met at present," and that it was working to increase capacity. CVS confirmed that manufacturers have been unable to provide sufficient supplies of several estrogen products.
Dr. Kathleen Jordan, chief medical officer at Midi Health, a telehealth company specializing in women's health, said the company has seen intermittent shortages in pockets throughout the U.S. since 2022. The current shortage is causing more disruption than previous episodes. Clinicians have been steering patients toward alternatives where possible, including oral estradiol pills, topical gels, and creams, all of which deliver the same hormone through different mechanisms. Telehealth services that focus on hormone therapy are connecting patients with menopause specialists who can prescribe those alternatives.
The shortage exposes a structural flaw that extends beyond manufacturing logistics. Medical organizations openly acknowledged that removing the black box warning would increase interest in hormone therapy. That was the goal. Yet production capacity did not expand in anticipation of that shift, and no parallel investment was made to ensure supply would meet renewed demand. The American Society of Health-System Pharmacists lists multiple estrogen products with current or recent shortages, and the disruption is expected to persist through the end of 2026. For the millions of women who went untreated during two decades of overstated warnings, the wait for a stable supply is its own kind of injustice.
This article was produced by Prism’s automated news system from verified source data, official records, and press releases, then run through automated quality and moderation checks before publishing. The system is built and supervised by the people who set the standards it runs under. Read our full AI policy.
Know something we missed? Have a correction or additional information?
Submit a Tip

