Johnson & Johnson says AI cuts drug lead development time in half
Johnson & Johnson says AI has cut drug lead optimization time by 50%, but the company still says clinical testing and regulation will take years.

Johnson & Johnson says artificial intelligence has cut by half the time its scientists need to optimize drug leads, a gain that speeds the front end of discovery but does not shorten the long path through clinical trials, regulation and manufacturing. Jim Swanson, the company’s executive vice president and chief information officer, delivered the update at Reuters Momentum AI New York 2026 in New York.
Swanson said Johnson & Johnson is using AI to screen the “potential universe” of chemical compounds and biologics for promising candidates. The company says traditional drug discovery takes years because researchers must sift through huge amounts of data across vast chemical space to find molecules with the right chemical and biological properties. Its AI and generative modeling tools, by contrast, can evaluate millions of compounds in parallel. That may materially reduce waste in the earliest stage of research, but it is still only the first filter. The hard parts that determine whether patients ever benefit, including preclinical testing, human trials, regulatory review and scale-up, still take years.
“We’re trying to cure cancer,” Swanson said, adding that the company needs every tool it can leverage and that “We’ve cut our lead optimization time in half.” The wording matters: lead optimization is not the same as inventing a finished medicine. It is the stage where researchers refine the most promising compounds, improve their properties and decide which ones deserve the expensive attention that follows.

Johnson & Johnson’s own materials show how large the operation is behind that claim. Its Discovery, Product Development & Supply team includes about 4,000 drug hunters, drug developers and clinical supply experts. The company says that group functions as its molecular invention, development and delivery engine. In other words, AI is being slotted into an already massive industrial process, not replacing it.
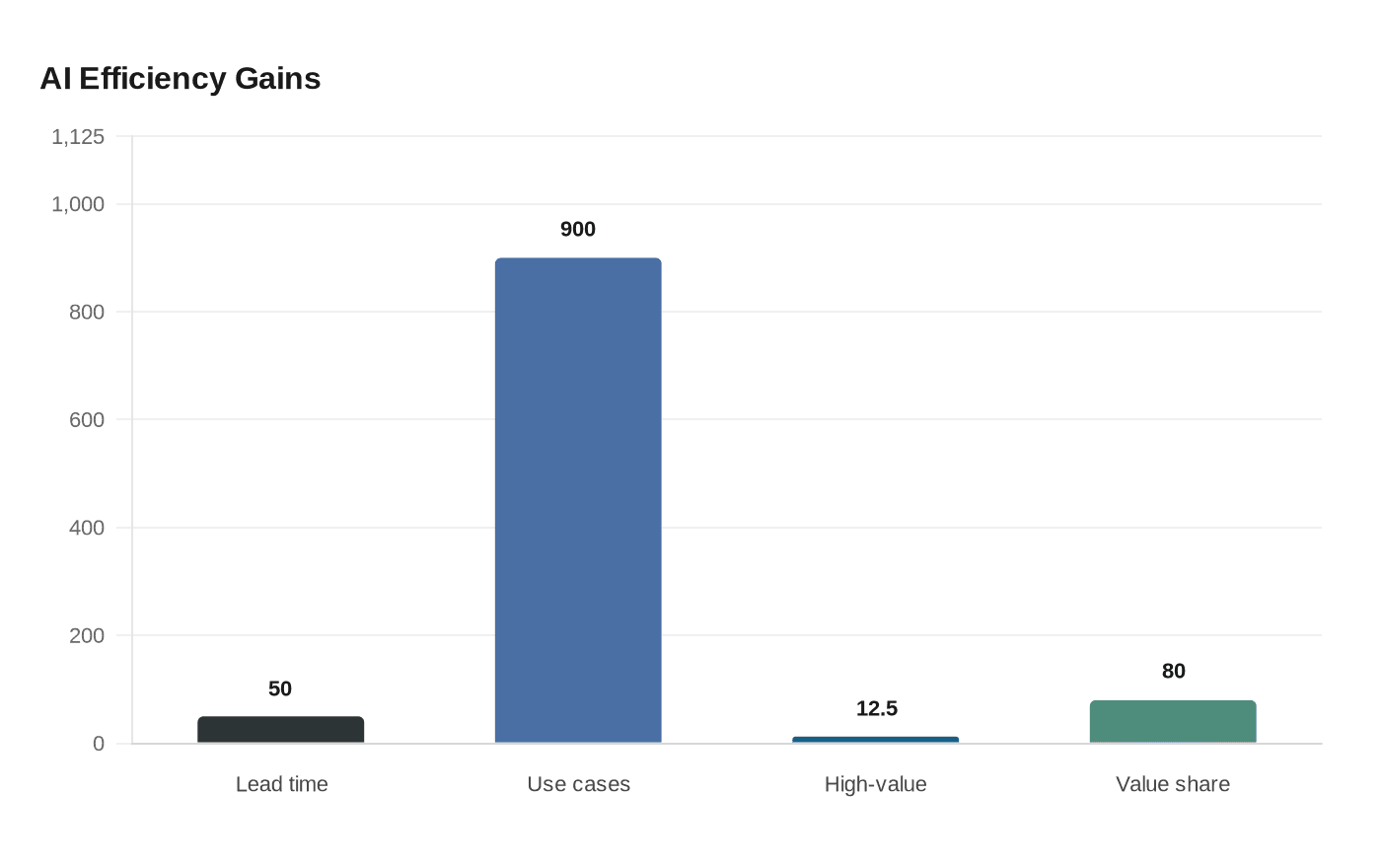
The company has also moved from experimentation to concentration. After testing nearly 900 generative AI use cases, Johnson & Johnson said only about 10% to 15% of them accounted for roughly 80% of the value, prompting a narrower focus on higher-return applications. Swanson has described the company’s AI deployment in three layers: enterprise productivity, end-to-end business processes such as drug discovery and clinical operations, and embedding AI into products and services.
That is the broader economic story here. Drug discovery remains one of the costliest and slowest parts of health care, and even a meaningful reduction in early-stage screening time can change the economics of research. But Johnson & Johnson is also making a more cautious argument: AI is best used as a faster, more disciplined filter inside regulated workflows where accuracy, traceability and governance matter as much as speed. The company says it has used data science and intelligent automation for more than a decade, and its latest message is that the payoff now lies in sharper efficiency, not a fully autonomous drug-making machine.
Know something we missed? Have a correction or additional information?
Submit a Tip

