Moderna to pay up to $2.25 billion to settle LNP patent fight with Genevant and Arbutus
Moderna will pay $950 million in July 2026 and could owe $1.3 billion more, resolving global litigation over lipid nanoparticle delivery used in its COVID-19 vaccines.
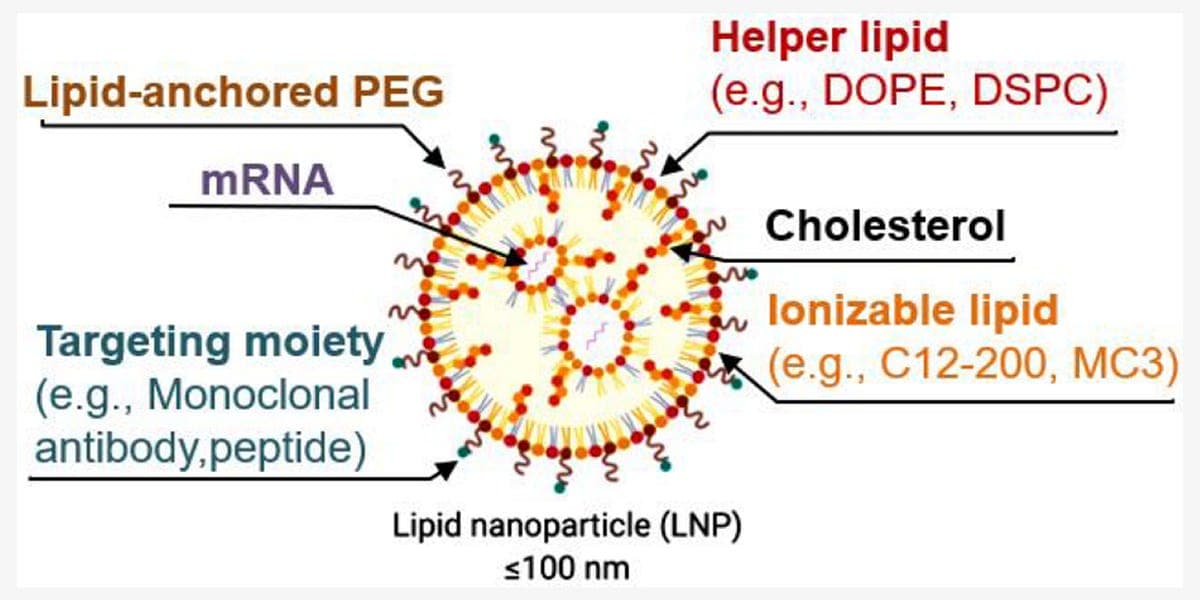
Moderna has agreed to a settlement that will require a $950 million upfront payment in July 2026 and could add up to $1.3 billion more, for a total potential payout of $2.25 billion, to resolve U.S. and international patent litigation with Genevant Sciences and Arbutus Biopharma over lipid nanoparticle delivery technology used in its COVID-19 vaccines, including Spikevax, the companies announced.
The joint GlobeNewswire press release, issued from Basel and Vancouver, and corroborating market feeds describe a package that combines cash, licensing and legal concessions. Genevant, a Roivant Sciences subsidiary described in the announcement as a leading nucleic acid delivery company, will grant Moderna a global non-exclusive license to its LNP delivery technology for infectious disease applications. The settlement also includes a covenant not to sue on certain patents and resolves “all U.S. and international enforcement actions” tied to the companies’ allegations of unauthorized use.
The agreement contains a significant legal admission: Moderna has consented to entry of a judgment of infringement and of no invalidity for four Genevant/Arbutus patents, according to the companies’ statement. The firms directed readers to Arbutus’ Form 8-K filed with the SEC on March 3 for additional details on the settlement terms and contingent payment mechanics.
The additional $1.3 billion is contingent on an appellate ruling concerning 28 U.S.C. § 1498, commonly referred to as Section 1498. Under that provision, the U.S. government can be treated as a defendant for patent claims involving government-contracted supplies. The contingent payment will be owed only if an appellate court rules that Section 1498 does not bar Genevant’s and Arbutus’ claims against Moderna for patent infringement, except as to doses the district court characterized as having gone to U.S. government employees. Market feeds noted that Moderna had argued Section 1498 shields certain government-contracted sales from patent liability.

The settlement carries immediate financial and policy implications. A $950 million payment scheduled for next July is large enough to affect Moderna’s short-term cash planning and investor calculations, while the $1.3 billion contingency puts a high-stakes legal question at the center of whether patent claims tied to government procurement are compensable. For public health and equity advocates, the carve-out tied to government-employee doses raises questions about how taxpayer-funded purchases and emergency use logistics intersect with intellectual property rights.
Beyond balance sheets, the deal changes the landscape for LNP technology licensing. A global non-exclusive license limited to infectious disease applications preserves the ability of Genevant and Arbutus to commercialize or license the technology in other fields, but it also gives Moderna a clear pathway to use LNP technology in future infectious disease products without further litigation tied to the settled patents.
Reporters and stakeholders should review Arbutus’ March 3 Form 8-K for the settlement agreement and patent identifiers to understand the scope of the admitted infringement and the licenses granted. The contingent payment will hinge on appellate timing and rulings that could take months or longer, leaving significant uncertainty around the final financial toll and the precedents it may set for government-contracted biomedical products and industry licensing practices.
Know something we missed? Have a correction or additional information?
Submit a Tip

