Modified CAR-T therapy may spare blood cancer patients toxic chemotherapy
A stem-cell memory CAR-T approach produced 45% complete responses without pre-treatment chemotherapy, hinting at a gentler path for hard-to-treat blood cancers.
A modified CAR-T therapy built from stem-cell memory T cells delivered complete remissions in a small first-in-human study without the harsh chemotherapy that normally prepares patients for treatment, a result that could make the cancer therapy easier to tolerate for some people with advanced blood cancers.
The study, published in Cell, tested a CAR-T product made from stem-cell memory T cells, or TSCM, a rare T-cell subset prized for self-renewal and long-term persistence. Patients in the trial had a range of blood cancers and had already failed bone marrow transplant treatment. No participant received chemotherapy before infusion, a striking departure from standard CAR-T practice.
Results were mixed but encouraging. Complete response rates were 45% in the TSCM group versus 10% in the standard CAR-T cohort, while overall response rates were statistically similar. One report said the standard cohort received a median dose of 290 million modified cells, compared with 66 million in the memory-stem-cell group. Another highlighted responses at doses as low as 250,000 cells per kilogram.
The modified cells also appeared to persist longer. Researchers reported a median time to adverse events or disease progression of 3.3 months in the standard CAR-T cohort and 4.9 months in the CAR-T stem-cell cohort. Four TSCM recipients went more than two years without disease progression. The memory-stem-cell group also showed lower rates of a common inflammatory reaction tied to overactive CAR-T cells, one of the best-known risks of the treatment.
Luca Gattinoni of the Leibniz Institute for Immunotherapy in Regensburg, Germany, led the study, with James Kochenderfer of the National Cancer Institute as co-corresponding author. Gattinoni said the findings open a new chapter in CAR-T design. Institutional coverage from the Leibniz institute said the results validate years of preclinical work, while stressing that the approach remains early.
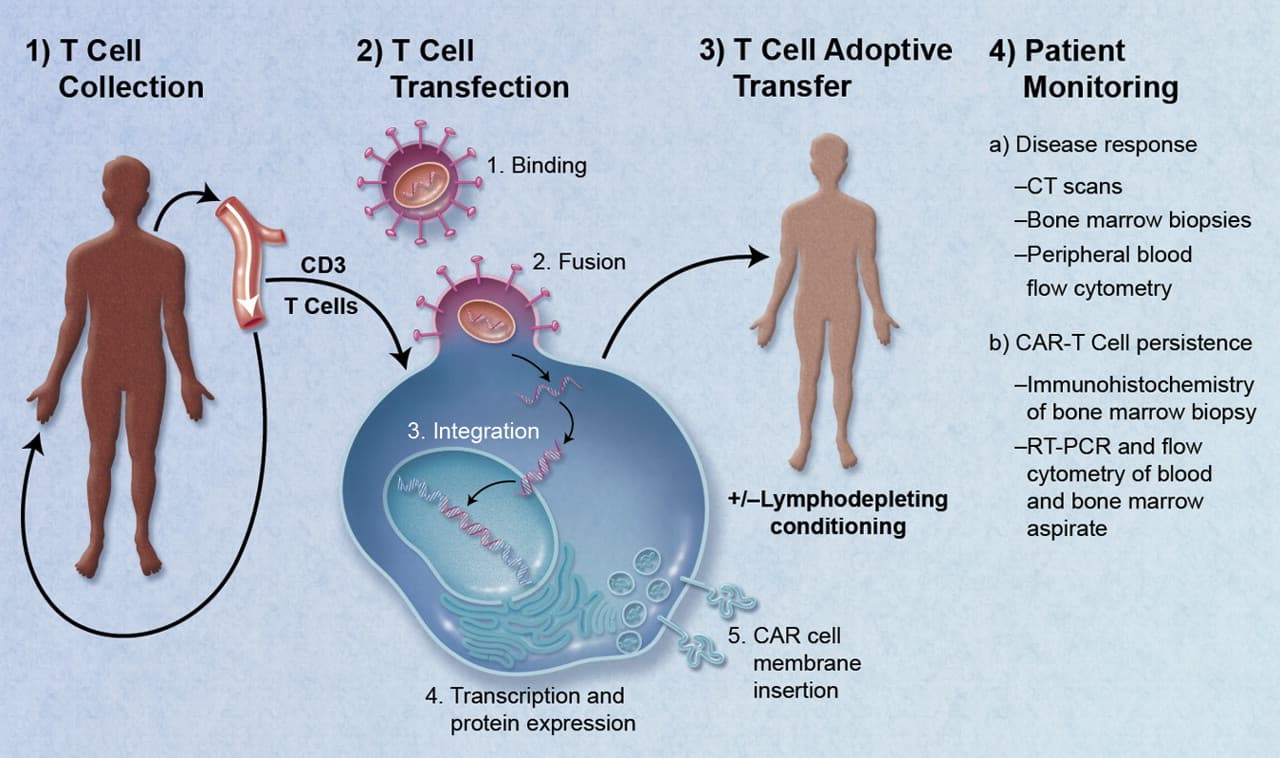
That caution matters. CAR-T has become part of modern cancer care over the past decade, but it still does not work for everyone. The Food and Drug Administration first approved a CAR-T therapy in 2017 for children with acute lymphoblastic leukemia, and later approvals expanded to adult blood cancers including non-Hodgkin lymphoma and multiple myeloma. The treatment uses a patient’s own T cells, engineers them to recognize cancer and returns them to the body as what the National Cancer Institute has called a “living drug.”
If larger studies confirm the early signal, the biggest payoff may be practical as much as scientific. Avoiding or reducing pre-treatment chemotherapy could lower complications, shorten preparation, and broaden access for frailer patients who have already been through extensive therapy. For now, the new data suggest a path toward a less punishing version of one of oncology’s most powerful tools.
Know something we missed? Have a correction or additional information?
Submit a Tip