Personalized mRNA pancreatic vaccine shows lasting survival in early trial
Nearly all responders to a personalized mRNA pancreatic vaccine were still alive six years later, a striking signal in a 16-patient early trial.
Nearly all of the pancreatic cancer patients who mounted a response to a personalized mRNA vaccine were still alive six years later, a rare outcome in a disease that is usually caught late and remains notoriously hard to treat. The result came from a small phase 1 study of 16 people with resected pancreatic ductal adenocarcinoma and offers an early, but important, sign that a tailor-made immune strategy may help keep the cancer from returning.
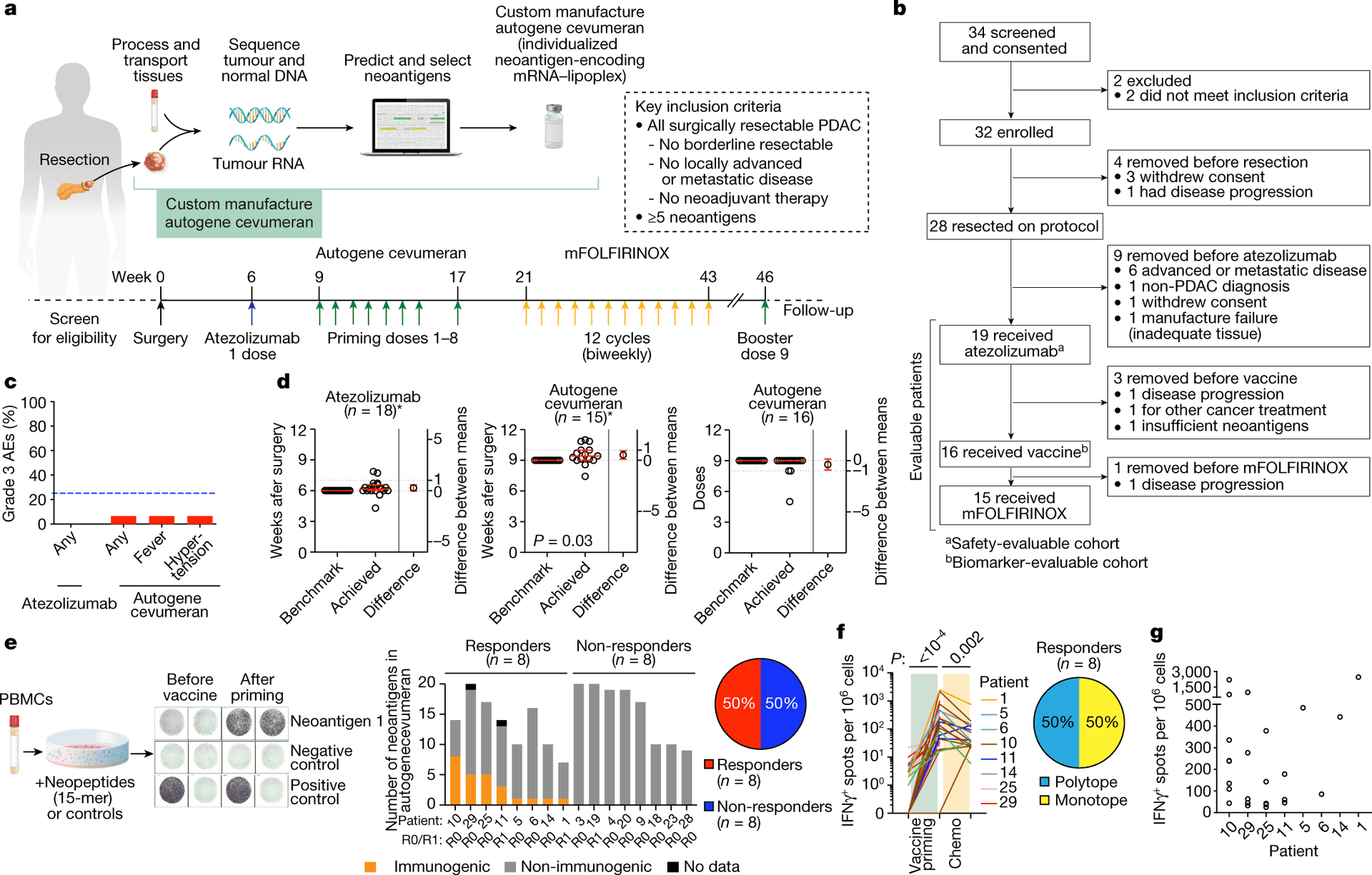
The vaccine, autogene cevumeran, also known as BNT122 and RO7198457, was developed by BioNTech and Genentech and given after surgery together with atezolizumab and modified FOLFIRINOX chemotherapy. In the Nature report published Feb. 19, 2025, researchers led by Memorial Sloan Kettering Cancer Center said eight patients had a vaccine-induced T-cell response. At a median follow-up of 3.2 years, those responders had recurrence-free survival that had not yet been reached, compared with 13.4 months in non-responders.

The immune response also appeared durable. The study found that vaccine-induced CD8+ T-cell clones had an average estimated lifespan of 7.7 years, with some predicted to last for decades. About 86% of the vaccine-induced clones per patient were still present at substantial levels roughly three years after vaccination, and in some patients those tumor-specific immune cells were detectable for nearly four years after treatment.
Memorial Sloan Kettering said the vaccine activated tumor-specific immune cells that persisted in the body for nearly four years in some patients. Outside experts have called the findings encouraging, but they also stressed that the work remains proof-of-concept. The central question is not whether the immune signal is real, it is whether that signal can be reproduced in larger groups and translated into better survival for more patients.
That next test is already underway. A randomized phase 2 study, NCT05968326, is comparing adjuvant autogene cevumeran plus atezolizumab and modified FOLFIRINOX against modified FOLFIRINOX alone in patients with resected pancreatic ductal adenocarcinoma. BioNTech said in 2023 that the trial was expected to enroll about 260 patients across the United States, Europe and Asia-Pacific.
For pancreatic cancer, where recurrence after surgery is common and long-term survival is still too rare, the prospect of a personalized mRNA vaccine offers cautious hope. The six-year survival signal is striking, but it remains an early result from a small trial, not a new standard of care.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip