Studies show CDC delay of infant hepatitis B shot may raise infections
Even a short delay in the hepatitis B birth dose could cost up to $370 million and leave more newborns infected. About 90% of infected infants develop chronic disease.

Delaying the hepatitis B shot at birth could add newborn infections and drive avoidable costs as high as $370 million, according to a Cornell University model of 3.6 million U.S. infants born in 2024. The study, published in JAMA Pediatrics, found that the later the first dose is given and the less reliably families return for follow-up shots, the higher the health and economic toll.
That warning lands as federal policy has moved away from a universal birth-dose recommendation for babies whose mothers test negative for hepatitis B surface antigen. In December 2025, the Advisory Committee on Immunization Practices voted 8 to 3 for individual-based decision-making, and the Centers for Disease Control and Prevention adopted that approach on December 16, 2025. The CDC still recommends a birth dose within 12 hours of delivery, along with hepatitis B immune globulin, for infants whose mothers test positive for hepatitis B or whose status is unknown.
The stakes are larger than a scheduling change. About 90% of infants infected at birth go on to develop chronic hepatitis B, a lifelong infection that can lead to serious liver disease. Public-health sources say childhood and adolescent hepatitis B infections fell by about 99% after the universal newborn vaccine policy began in 1991, which had replaced an era when about 18,000 children a year were infected before age 10 and roughly half those infections occurred at birth.

The CDC’s own prior guidance has noted that, before universal birth-dose vaccination, some U.S.-born children of immigrant mothers without hepatitis B infection still had prevalence rates of 7% to 11% because of household or community exposure. That history is why public-health experts have argued that waiting for later vaccination leaves a gap in protection even when mothers screen negative during pregnancy.
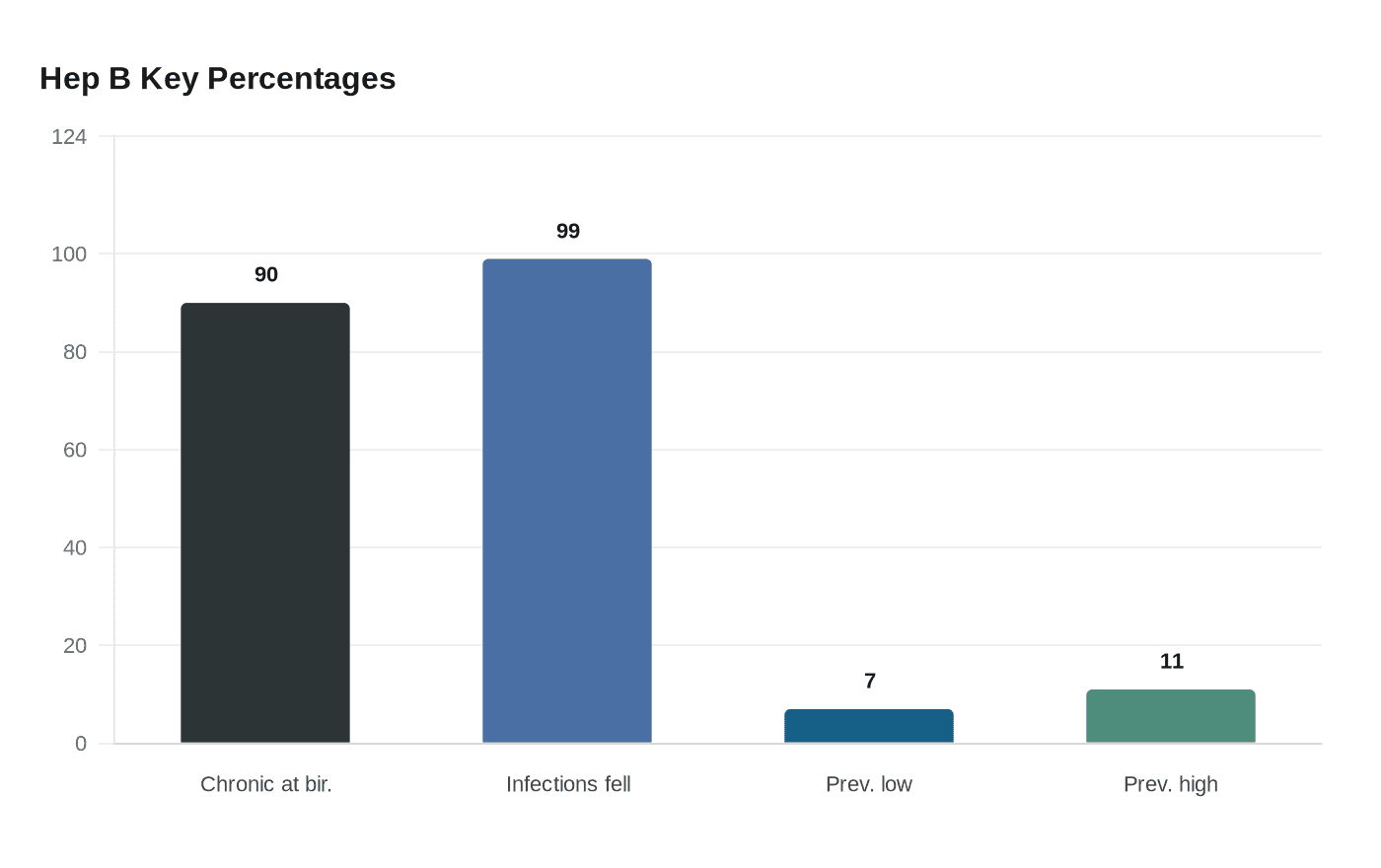
The American Academy of Pediatrics said after the December vote that it would continue recommending a birth dose, calling the change harmful to children. Other groups, including the American Public Health Association and researchers at Johns Hopkins Bloomberg School of Public Health, have warned that newborns remain vulnerable to exposure outside the delivery room. The economic case is equally stark: Cornell’s model projected delay-related costs from $16 million to $370 million, a range that reflects how much more expensive prevention becomes once the first shot is pushed back.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip

