Takeda immune disease drug meets trial goal, cuts infusion burden
Takeda’s TAK-881 matched HyQvia’s antibody protection while using half the infusion volume, a change that could ease life for patients with primary immunodeficiency.
Takeda said its experimental primary immunodeficiency treatment, TAK-881, met the main goal of a mid- to late-stage trial by producing protective antibody levels comparable to its approved product HyQvia. For patients who depend on recurring plasma-derived infusions, that result matters because the new drug did more than match the blood-marker target: Takeda said it used half the infusion volume and took less time to administer.
That combination could make a real difference for people living with rare immune disorders, especially children and families who already spend hours in infusion chairs or at home managing repeated treatment sessions. The company said the study included adults and children as young as 2 years old who were already receiving immunoglobulin therapy. It also said TAK-881 had a safety profile similar to HyQvia, with no new safety concerns identified, which is important in a disease area where long-term tolerability can shape whether patients stay on therapy.
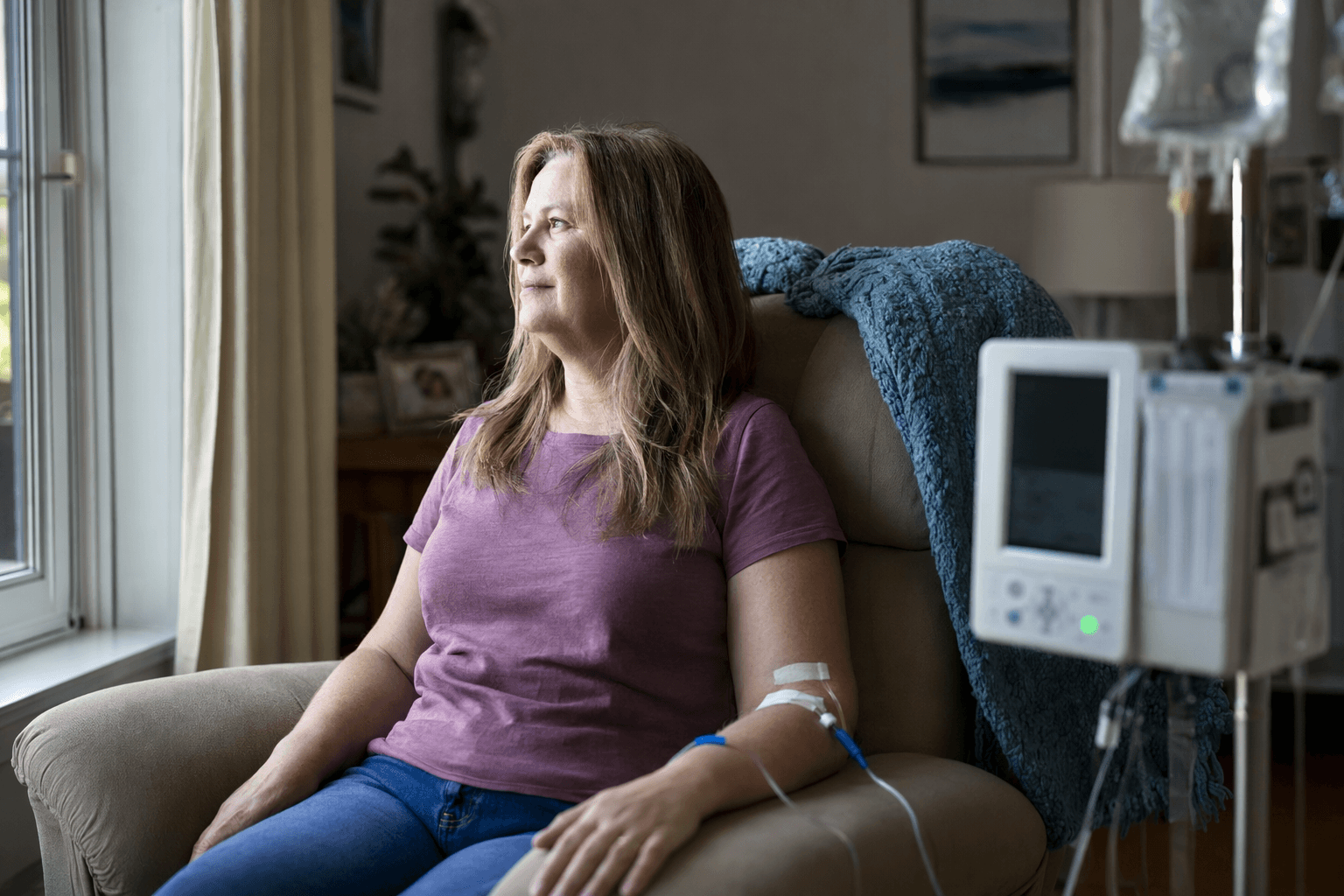
Primary immunodeficiency is not just a laboratory problem. It is a daily burden that can disrupt school, work and family routines when treatment is frequent and lengthy. In that setting, a therapy that maintains the same level of immune support while shrinking infusion volume and shortening administration time could be more than a modest convenience improvement. It could reduce the friction that keeps patients from sticking with treatment, while preserving the antibody protection they need to help guard against infection.

Takeda said it plans to seek approval in the United States, the European Union and Japan in fiscal 2026. That means the trial result is a step toward possible use in the clinic, not an immediate change in care. If regulators accept the data, TAK-881 would enter a market where convenience, safety and reliability matter almost as much as efficacy, and where even small reductions in treatment burden can have outsized effects on quality of life. For people with primary immunodeficiency, that is the central question: not whether the drug works in a lab sense, but whether it can deliver the same protection with less of a toll on everyday life.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip