Toxic Ink commentary sparks renewed concerns over tattoo pigment migration, lymph-node effects
Lab testing of 15 tattoo inks found eight EU‑restricted metals and titanium up to about 10,000 ppm in a light-blue ink, while a March 7, 2026 medical essay framed ink as a systemic risk.

Laboratory analyses of 15 tattoo‑ink samples detected a suite of regulated toxic metals and raised fresh questions about pigment migration and lymph‑node accumulation. The analytical work, described as using “advanced analytical techniques,” identified eight metals that are restricted under EU law: antimony, arsenic, cadmium, chromium, copper, lead, selenium and tin, and found that at least one ink exceeded limits set under EU Commission Regulation 2020/2081 and the Council of Europe Resolution ResAP (2008).
The report singled out non‑restricted additives used for colour and stability: “Titanium was detected at concentrations of up to about 10,000 parts per million in a light-blue ink,” Dr Violi says. Dr Violi also said, “Aluminium and zirconium were also present at very high levels. These metals are commonly associated with pigments that are used to improve colour and stability.” The presence of those elements at high concentrations prompted researchers to note that pigments can persist in the skin and migrate to lymph nodes, a pathway that creates toxicological questions when metals are present.
The analytical findings appeared as the debate over ink safety intensified after a March 7, 2026 long‑form medical‑opinion essay by Dr. Robert Yoho, MD, framed tattoo ink as a systemic health risk and surveyed literature on pigment migration and lymph‑node effects. The essay’s framing contrasts with the laboratory team’s more measured public language: “However, the researchers say their findings should not be interpreted as evidence that tattoos directly cause harm.”
Beyond the single light‑blue ink with elevated titanium, the testing flagged regulatory non‑compliance more broadly. The reporting states, “Across the world, investigations document widespread non-compliance with EU standards. This indicates tattoo inks remain a significant and under-regulated source of toxic metal exposure.” The combination of exceedances under EU Commission Regulation 2020/2081 and the Council of Europe Resolution ResAP (2008) puts regulatory limits squarely into focus for artists and suppliers who import pigments or buy ready‑made inks.
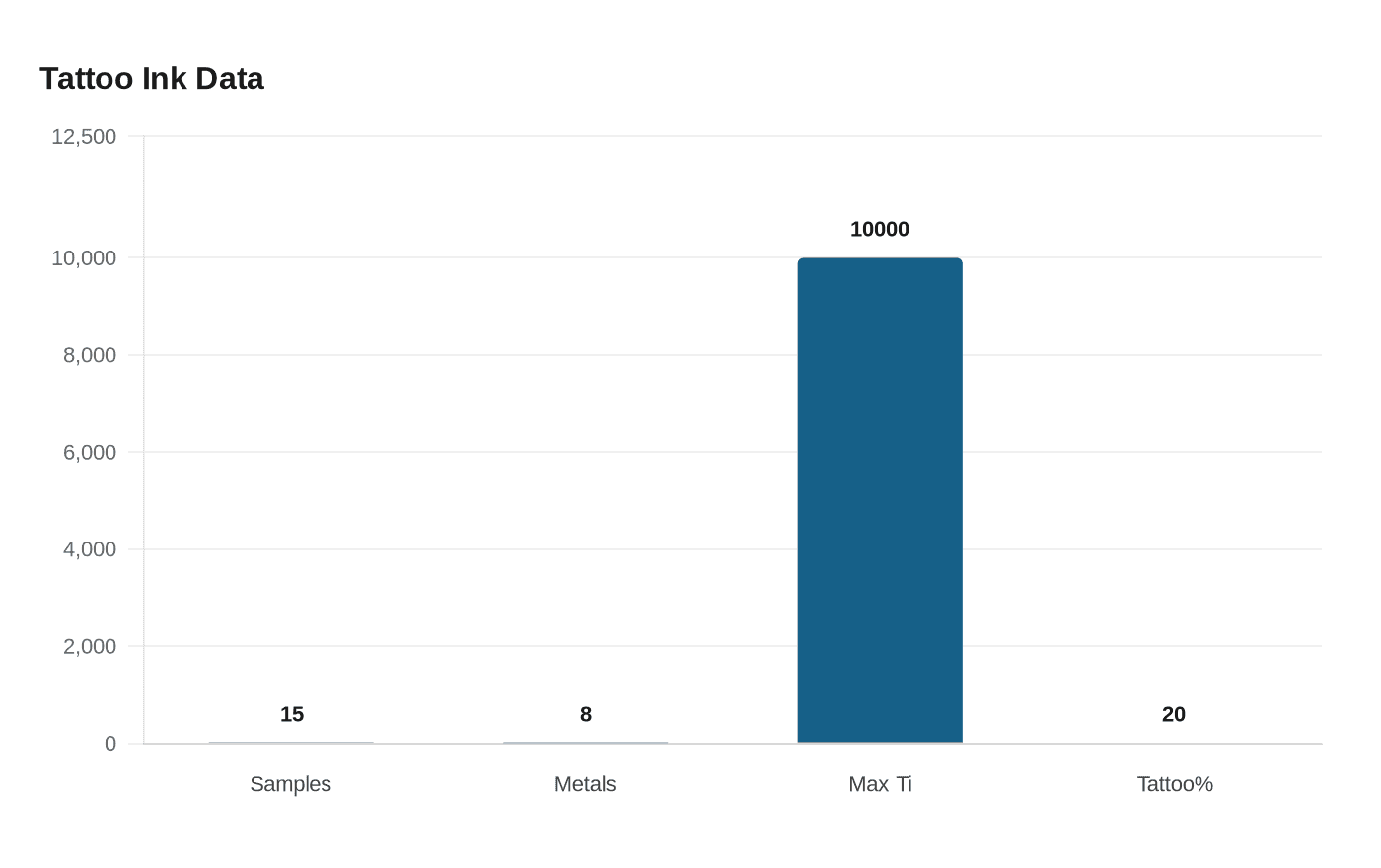
The findings also re‑energize questions raised in Australia, where surveys estimate more than 20 percent of adults have at least one tattoo. Australia’s only government survey of tattoo inks, conducted in 2016 with an update in 2018, concluded that most inks tested would not meet European guidelines, though the government report included limited methodological detail and predated the EU’s current legally binding rules.
Important gaps remain: the testing covered 15 samples with no public full dataset in the supplied material, the identities and affiliations of the laboratory authors including Dr Violi were not provided, and the specific concentrations for the EU‑restricted metals beyond the single titanium figure were not detailed. The exceedances under Regulation 2020/2081 and ResAP (2008), combined with the persistence and migration concerns raised by researchers and by Dr. Robert Yoho’s survey of the literature, point to an urgent need for transparent datasets, clearer chain‑of‑custody in testing, and regulatory review of pigment supply chains and enforcement.
Know something we missed? Have a correction or additional information?
Submit a Tip

