WHO urges tighter rules on nicotine pouches amid youth marketing push
Nicotine pouch sales topped 23 billion units last year as WHO warned flavors, influencers and sponsorships are pulling in young users. Only 32 countries regulate the products.
The World Health Organization is pressing governments to tighten nicotine-pouch rules as the products spread faster than laws can catch up, warning that colorful packaging, flavors and online promotion are drawing in young users. The agency said retail sales topped 23 billion units in 2024, more than 50% above the previous year, and estimated the global market at nearly US$7 billion in 2025.
In its first global report on nicotine pouches, WHO said regulation is limited or absent in around 160 countries. Just 16 countries ban sales outright, while 32 regulate the products in some form. Among those, only five restrict flavors, 26 restrict sales to minors and 21 ban advertising, promotion and sponsorship, leaving a wide regulatory gap for a category that is being sold as a modern, cleaner alternative to smoking.

Etienne Krug, who directs the WHO Department of Health Determinants, Promotion and Prevention, said, “These products are engineered for addiction.” WHO said some pouch brands are marketed as “beginners,” “advanced” and “experts,” and that nicotine concentrations can reach as high as 150 milligrams. The agency said the products usually contain nicotine, flavorings, sweeteners and other additives, and warned that nicotine exposure in adolescence can affect attention and learning, increase the likelihood of long-term dependence and future use of other nicotine and tobacco products, and is associated with increased cardiovascular risk.
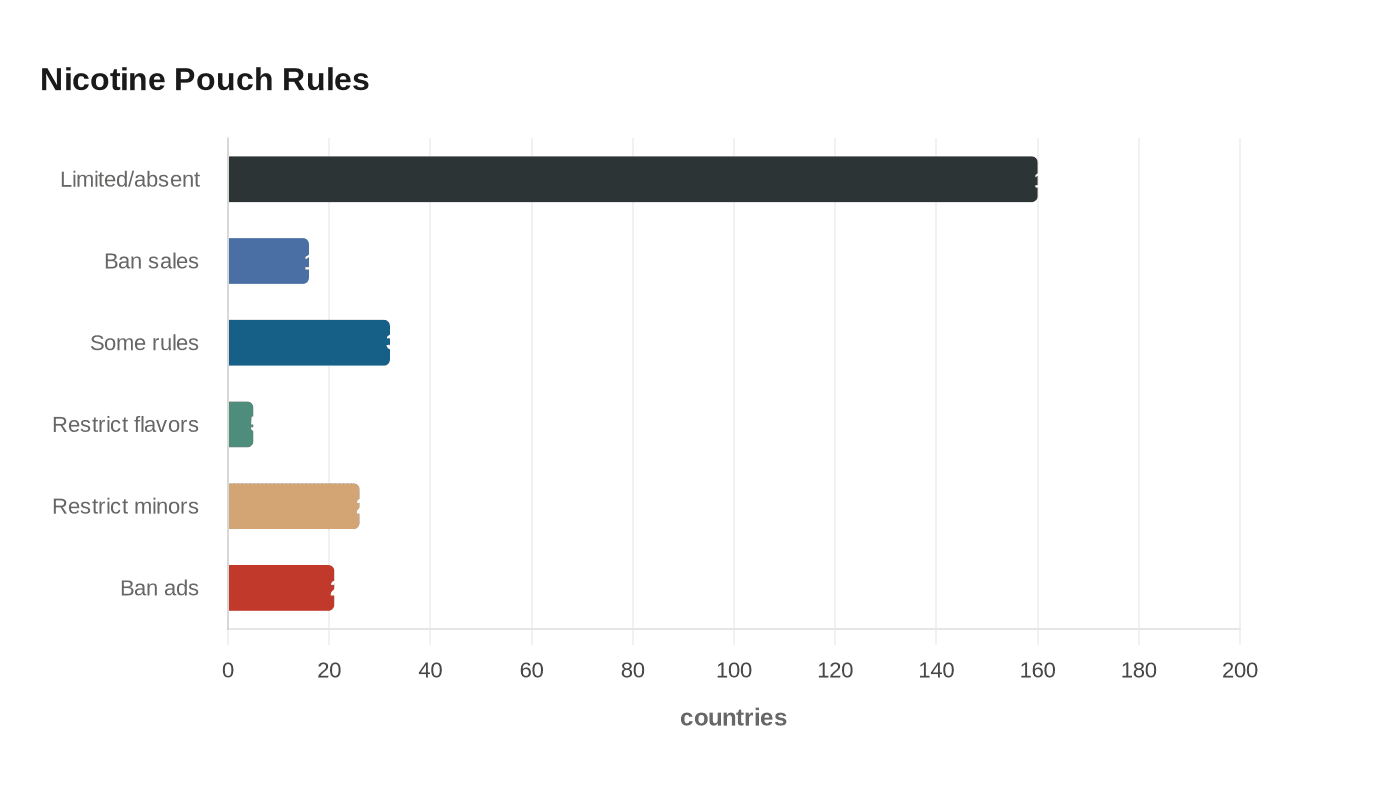
The warning lands as nicotine pouches, including Zyn, are now sold in dozens of countries and have become one of the tobacco industry’s fastest-growing product lines as cigarette consumption declines. Public-health advocates say the marketing echoes the youth-vaping playbook, with social media influencers, music festivals, concerts and sports sponsorships helping normalize the products. The American Lung Association has argued that flavored pouch authorization can benefit the tobacco industry and could encourage youth-focused marketing, keeping the policy fight centered on whether the products are harm-reduction tools for adult smokers or a new addiction vector for teenagers.
In the United States, the Food and Drug Administration issued a filing letter on June 17, 2025, for ZYN modified-risk tobacco product applications, beginning scientific review. WHO said its report was developed in response to requests from countries seeking guidance on how to respond to the product category, and it comes ahead of World No Tobacco Day on May 31, when the 2026 theme will focus on tobacco and nicotine addiction and the industry tactics used to hook a new generation of users.
Know something we missed? Have a correction or additional information?
Submit a Tip