Wim Hof Method Reduces MS Inflammation Markers in 12-Week Pilot Study
43 MS patients showed drops in cytokines IL-17A and IFN-γ after 12 weeks of supervised Wim Hof breathing and cold immersion in a randomized pilot.

Forty-three people with multiple sclerosis completed 12 weeks of structured Wim Hof Method training in a randomized pilot, and the results moved a specific needle: blood levels of IL-17A, a cytokine tightly linked to MS-related chronic inflammation, fell in both the WHM and lifestyle intervention groups compared with controls. The WHM group went further still, recording an additional reduction in IFN-γ, the immune signaling protein that directly promotes attacks on the myelin sheath.
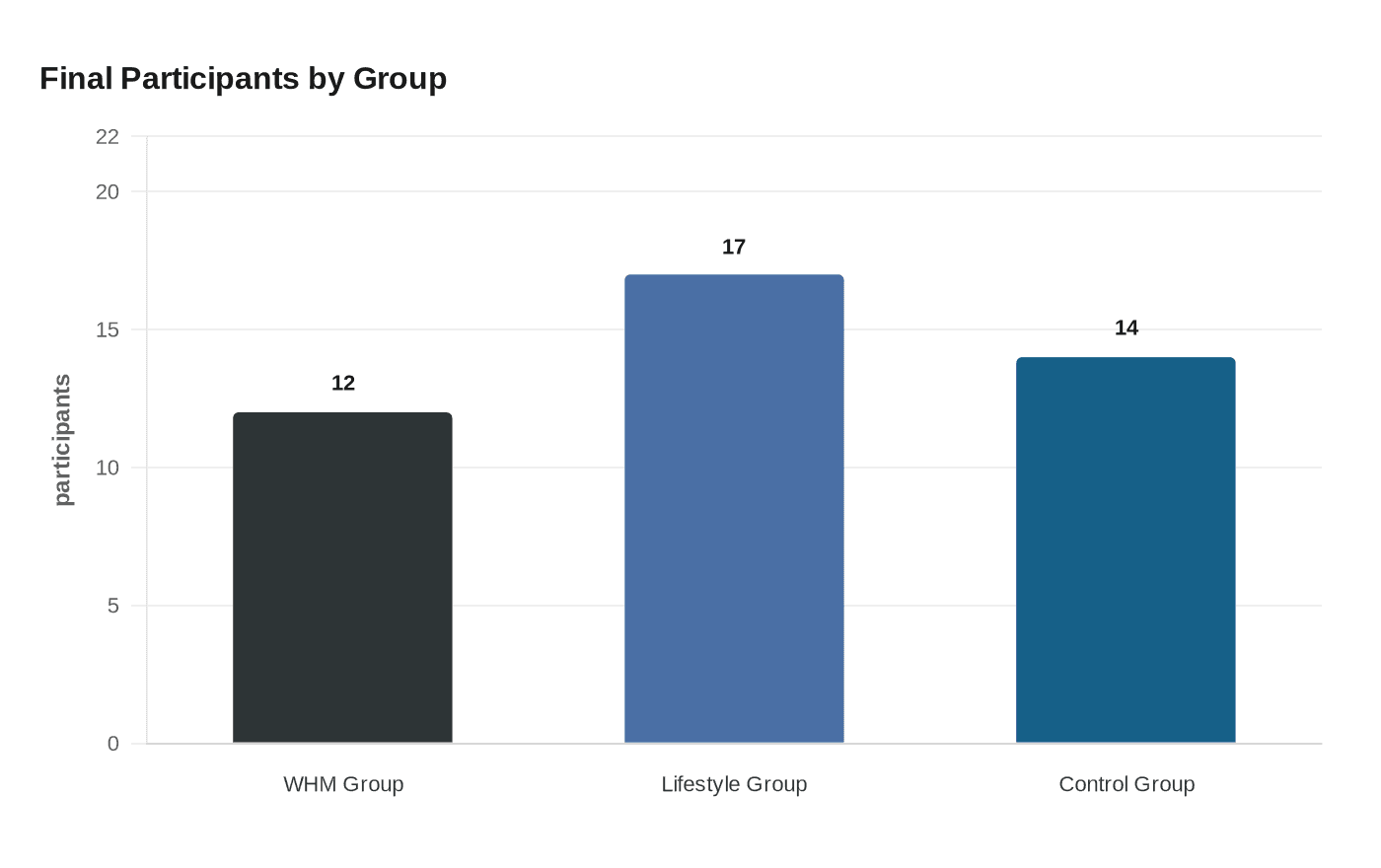
The study, "Targeting low-grade inflammation in multiple sclerosis through the Wim Hof method or lifestyle intervention: a pilot comparative study," published in Neurological Sciences and led by Darina Slezáková and colleagues out of Slovakia, enrolled 60 adults diagnosed with MS across three arms. Attrition left 12 in the WHM group, 17 in the lifestyle group, and 14 controls for the final analysis. Neither group achieved significant changes in neurodegeneration markers, and the small sample caps how confidently those null findings can be read.
The WHM protocol was specific. Each week, participants attended a supervised session running 40 to 55 minutes of guided breathing with focused attention and visualization, immediately followed by brief cold-water immersion and warming exercises. Between sessions, participants practiced structured breathing and cold exposure at home. The LIFE arm was comparably active: twice-weekly supervised dance-based exercise sessions paired with personalized reduced-calorie nutrition counseling. The control group received no intervention.

The investigators' own language reflects appropriate scope: "Both WHM and lifestyle modification demonstrated comparable short-term anti-inflammatory effects in MS, supporting their safety and feasibility as adjunctive strategies to DMT." The word "adjunctive" matters. Disease-modifying therapies control acute relapses more effectively than they suppress the slow, smouldering inflammation driving gradual nerve damage; neither protocol was studied as a DMT replacement, and the trial was not powered to measure relapse rates, disability progression, or functional outcomes.
Before replicating the protocol at home, five questions are worth raising directly with a neurologist or MS specialist. First, ask whether your disease is stable enough to safely tolerate repeated cold-water immersion, since autonomic stress from cold exposure is not benign for every MS presentation. Second, clarify whether your current DMT already targets IL-17A or IFN-γ pathways, which would affect how much additional anti-inflammatory benefit to expect. Third, ask whether a supervised WHM program is preferable to self-guided cold exposure given that every session in this trial was monitored by researchers. Fourth, establish what a realistic 12-week monitoring plan looks like for you before starting. Fifth, set a symptom baseline first, so any change is measurable rather than felt.
That last point connects directly to what a home tracker can realistically capture. Fatigue scores logged daily, sleep quality ratings, and a simple relapse symptom diary across 12 weeks produce data that placebo effects cannot convincingly sustain over time. The trial measured blood cytokines; that is not replicable at home. But tracking fatigue onset, sleep continuity, and any new or worsening neurological symptoms creates a personal record structurally similar to the monitoring this trial performed, and it gives any future clinical conversation something concrete to work from.
The Slezáková team's pilot is the first randomized study to test WHM against both an active comparator and a control group in a clinical MS population with biomarker endpoints. The feasibility data it generated now makes a strong case for larger trials with longer follow-up and functional outcome measures.
Know something we missed? Have a correction or additional information?
Submit a Tip

