Catalent cuts 96 jobs at two Baltimore-area facilities, shrinks Maryland workforce
Catalent filed a Feb. 19 WARN notice to cut roughly 96 roles across two Baltimore-area facilities, shrinking local gene-therapy staff after prior rounds that removed hundreds of jobs.

Catalent filed a WARN notice on Feb. 19 and confirmed to Technical.ly reporters Katie Malone and Danya Henninger that it will eliminate roughly 96 positions across two Baltimore-area facilities, part of another round of reductions affecting the company’s Maryland operations. Catalent manufactures the Duchenne muscular dystrophy therapy ELEVIDYS at its Maryland sites and said, “While these changes are difficult, our Baltimore operations and our gene therapy business remain critical to Catalent.”
The latest 96-job reduction follows three major moves in the past year: an August 2025 announcement that eliminated about 350 Maryland roles, a November 2025 round that cut 77 positions, and the Feb. 19 filing. Technical.ly and BioBuzz trace the August 2025 total to a sudden change in demand from a large commercial customer; Catalent’s spokesperson told Technical.ly, “Following last year’s announcement regarding a shift in demand from a large commercial customer, we have made additional, expected workforce reductions at our Baltimore site.”
Site-level WARN notices previously reported by FiercePharma break the November 2025 number into a Harmans site reduction of 61 workers with an effective date of Jan. 5 and another 16 employees across Baltimore and Hanover sites, totaling 77. The Baltimore Banner has published different figures for the August 2025 reductions, reporting 316 cuts at two Harmans gene-therapy facilities and 32 at the University of Maryland, Baltimore BioPark; those Banner numbers diverge from other public counts and remain part of shifting tallies cited across industry reporting.

Regulatory context adds to market pressure on Catalent’s Maryland operations. Citybiz reporter Alessandro Maselli noted a temporary FDA halt on ELEVIDYS shipments last month, with the agency later clearing limited distribution after reviewing reports of acute liver failure. Technical.ly also flagged a 2025 FDA inspection record that raised concerns about glove-sanitization practices and facility infrastructure at Maryland sites; those findings have been mentioned by industry outlets as background to the company’s restructuring.
The cuts come amid broader contraction in Maryland’s life-sciences cluster. BioBuzz’s internal layoff tracker lists more than 1,200 positions eliminated in Maryland in 2025, Citybiz cited state records showing more than 2,700 positions cut across multiple employers in May, and BioWorld figures quoted by BioBuzz put national biopharma reductions this year above 32,000. Other local moves include BioNTech’s disclosure of 63 layoffs in Gaithersburg and Eurofins’ 25 layoffs in Frederick, cited by Citybiz.
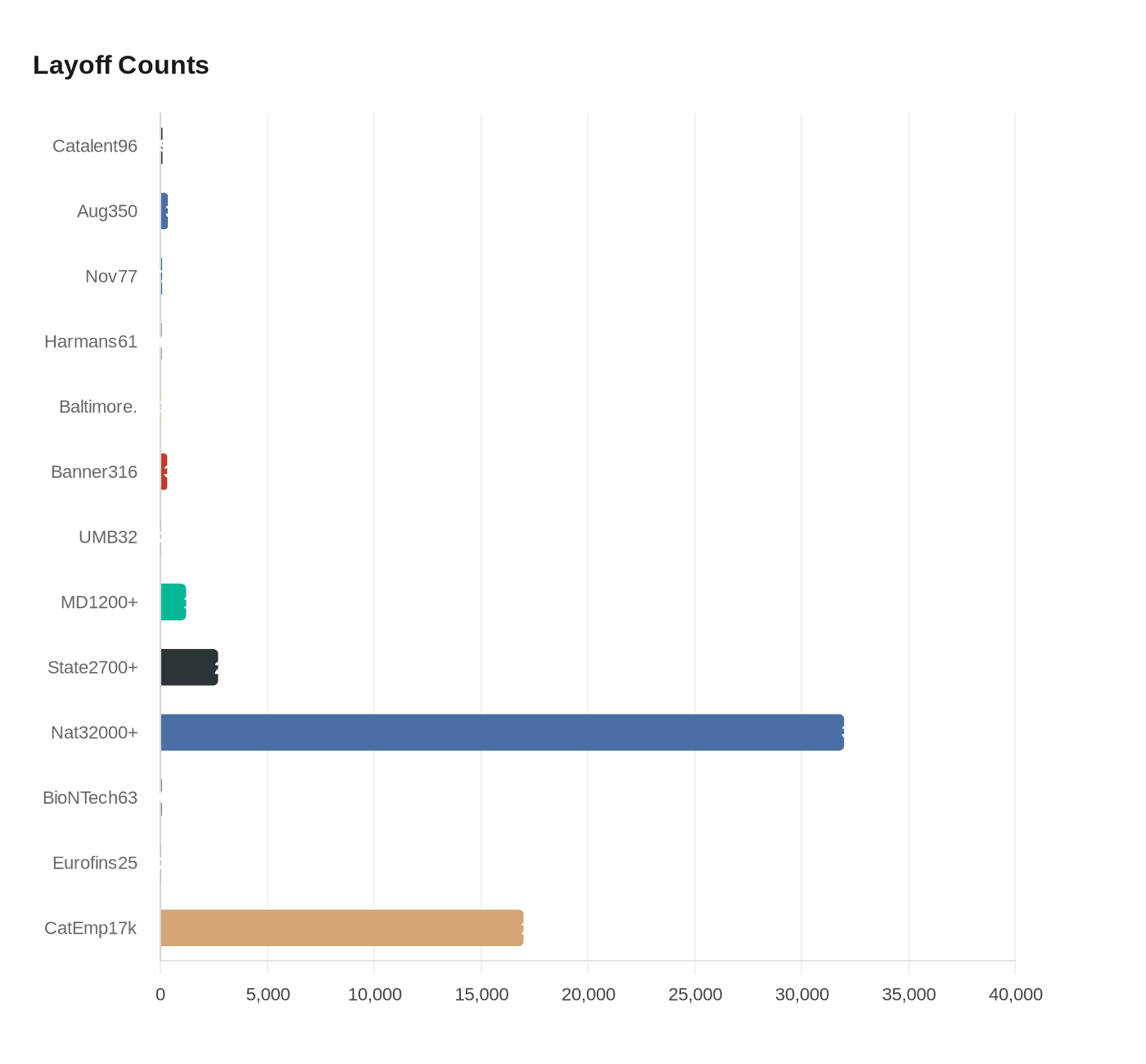
Catalent’s corporate footprint has also shifted during the last year: the company reports roughly 17,000 employees globally, opened a new corporate headquarters in Tampa in 2025 and has closed a Belgium site earlier this year, according to industry reports. FiercePharma noted an approaching anniversary of Catalent’s sale to Novo Holdings as part of broader corporate changes. Media reporting differs on the company’s described base, with some outlets characterizing Catalent as headquartered in Florida and others describing a New Jersey connection.
Catalent told BioBuzz it is providing transition support and remains “confident in our gene therapy business,” saying in a company statement, “It is never easy to see talented colleagues leave Catalent, and we remain committed to providing them transition support as they manage this change.” The Feb. 19 WARN and the successive reductions in August 2025 and November 2025 together mark another contraction for Baltimore-area gene-therapy operations at a moment when local employers and regulators are negotiating safety, demand and production challenges that have real implications for jobs and the region’s biotech cluster.
Know something we missed? Have a correction or additional information?
Submit a Tip

