Amgen Revenue Rises 6%, New Drugs Offset Patent Pressure
Amgen's revenue rose to $8.6 billion as 16 brands grew double digits, but investors still need proof that Tepezza, MariTide and other new bets can outlast patent pressure.
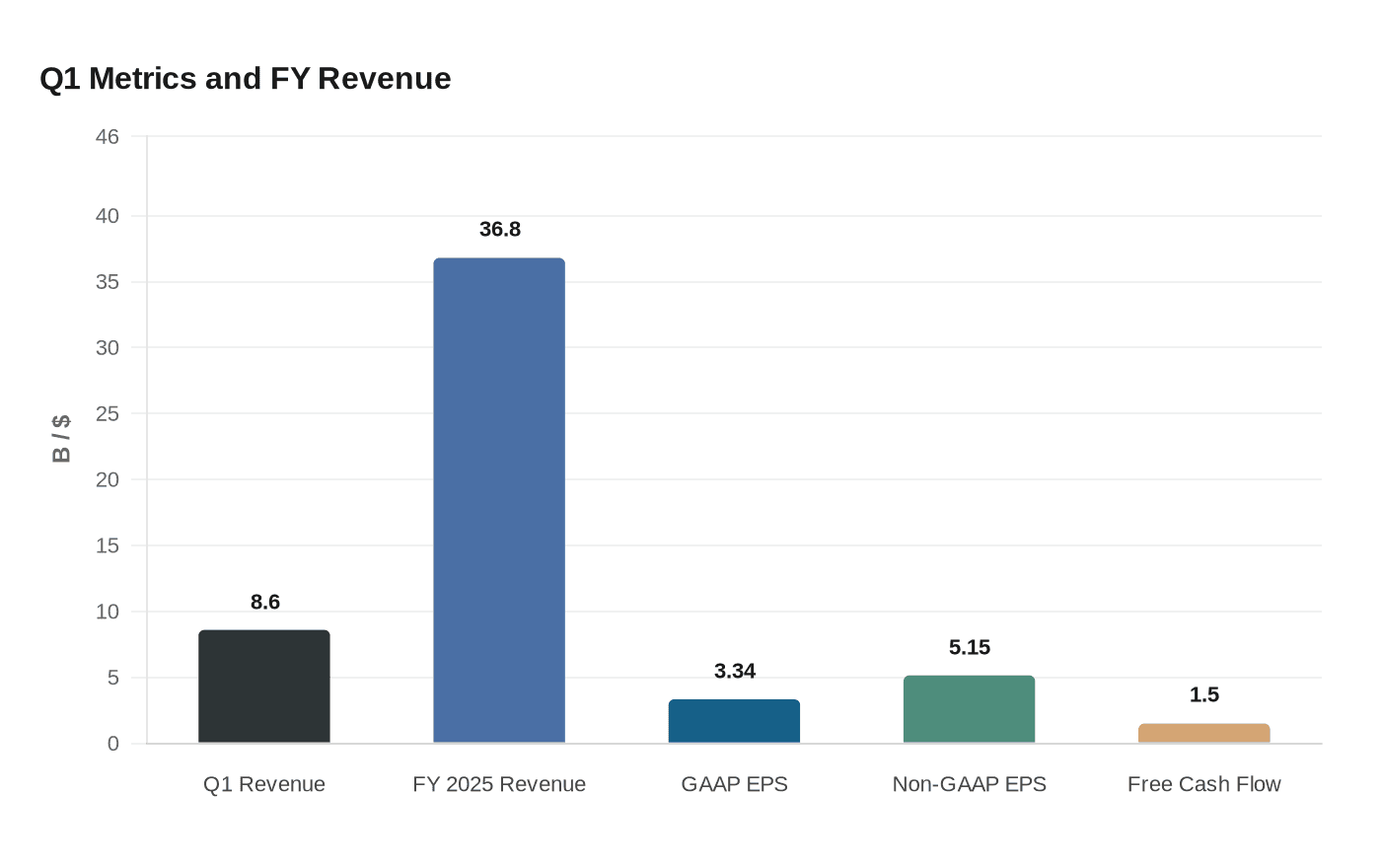
Amgen’s first-quarter revenue climbed 6% to $8.6 billion, a result that showed the company can still grow while older franchises face patent pressure and biosimilar competition. Product sales increased 4%, driven by 9% volume growth, even as lower net selling prices and lighter inventory levels trimmed some of that momentum. The company also posted a sharper earnings profile, with GAAP EPS rising to $3.34 from $3.20 a year earlier and non-GAAP EPS improving to $5.15 from $4.90.
The core message from Thousand Oaks, California, was not just that the quarter was strong, but that Amgen still has breadth in its portfolio. Chief Executive Robert A. Bradway said 16 brands delivered double-digit growth and that the company now has 17 products annualizing at more than $1 billion based on first-quarter sales. GAAP operating income nearly doubled to $2.7 billion, lifting the operating margin to 32.4%, while free cash flow rose to $1.5 billion from $1.0 billion. Those numbers suggest a business with enough scale to absorb pricing pressure for now.
That resilience matters because the more difficult question is what happens over the next two to three years. Amgen is leaning heavily on a new generation of products and late-stage candidates to extend the growth curve beyond mature blockbusters that will eventually lose exclusivity. Management said a new wave of molecules is advancing through Phase 3 development, a signal that the company wants investors to judge the quarter not only on current sales, but on whether the pipeline can replace future erosion fast enough.
The next catalysts are already visible. Amgen said on April 6 that subcutaneous Tepezza produced positive Phase 3 topline results in adults with moderate-to-severe active thyroid eye disease, while a separate April 3 update went to healthcare providers on Tavneos. That drug is under pressure after the U.S. Food and Drug Administration’s Center for Drug Evaluation and Research proposed withdrawing approval on April 27, citing concerns over effectiveness and the original application, even as Amgen said it was engaging with regulators and had submitted a label amendment to add more information on liver toxicity. Meanwhile, MariTide remains the company’s biggest long-term growth swing, with a Phase 3 cardiovascular outcomes study listed on ClinicalTrials.gov and an estimated enrollment of 12,800 participants.
Amgen entered 2026 with momentum after reporting full-year 2025 revenue of $36.8 billion, up 10%, and 18 products that reached record sales. The first quarter suggests that momentum has not broken, but it also shows the market’s central test: whether this mix of rising legacy brands and late-stage assets can bridge the company through the next patent cycle, or merely buy time.
Know something we missed? Have a correction or additional information?
Submit a Tip

