Autism Genetics Study Finds Risk Genes Shared Across Latin American Ancestries
Analyzing 15,000+ individuals, researchers identified 35 autism risk genes shared across Latin American and European ancestries, challenging genomics' historic European bias.
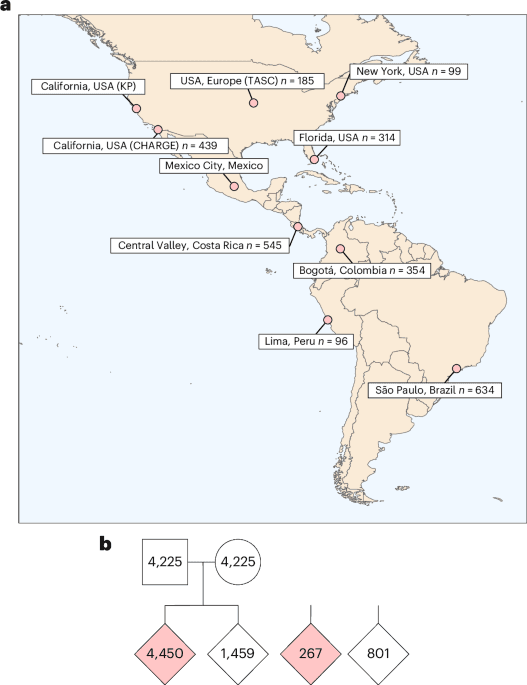
Autism's genetic risk factors appear to hold across human diversity. A study published March 30 in Nature Medicine, co-led by the Genomics of Autism in Latin American Ancestries (GALA) Consortium, identified 35 genome-wide significant autism-associated genes in a sequencing analysis of more than 15,000 individuals across the Americas, and found those genes overlap substantially with risk variants previously documented almost exclusively in European-ancestry populations.
The work represents one of the largest and most ancestrally diverse autism genomics datasets assembled to date. Researchers examined more than 18,000 genes for enrichment of rare, deleterious coding variants, drawing on roughly 4,700 participants diagnosed with autism spectrum disorder within the broader cohort of Latin American individuals. The analysis found that rare, damaging variants in highly conserved genes, those that perform essential biological functions across species, were disproportionately observed in autistic individuals, mirroring patterns established in prior European-ancestry studies.
Beyond reinforcing already-established autism genes, the study provided new statistical support for several emerging candidate genes, including MARK2, YWHAG, PACS1, RERE, SPEN, and GSE1. The authors concluded that the core genetic architecture of autism showed no detectable influence of ancestry in this large and diverse sample.
Joseph D. Buxbaum, PhD, director of the Seaver Autism Center for Research and Treatment at Mount Sinai and a senior author on the paper, put it plainly: "Our results indicate that the core genetic architecture of autism is shared across ancestries."
That conclusion carries significant weight in a field long dominated by data from European populations. Genomics research has faced sustained criticism for its Eurocentric sample bias, a structural gap with real clinical consequences. When reference datasets skew toward one ancestry, individuals from other backgrounds are more likely to receive inconclusive or ambiguous genetic test results. The GALA findings suggest clinicians ordering genetic panels for neurodevelopmental disorders can apply the same key risk genes across ancestries with greater confidence, though disparities in access to testing and local population variation remain ongoing concerns.

For families navigating an autism diagnosis, the practical promise is improved diagnostic clarity as diverse genetic reference data accumulates. For the broader scientific community, the conserved biological pathways implicated across populations point toward shared therapeutic targets, an important signal for drug and intervention research.
The study also makes a policy argument through its methods. Generating this level of cross-ancestry genetic insight required international consortium infrastructure, cross-border data sharing, and deliberate investment in recruiting participants from Latin America, a region characterized by recently admixed populations carrying distinct genomic signatures. Mount Sinai and affiliated institutions called for continued expansion of diverse participant recruitment, improved population-specific reference datasets, and broader availability of diagnostic and genetic counseling resources across underserved regions.
The GALA Consortium's work makes visible what precision medicine can achieve when the starting dataset actually reflects human diversity rather than assuming one population's biology speaks for everyone else's.
Know something we missed? Have a correction or additional information?
Submit a Tip

