Big Pharma Eyes Inhibrx Cancer Drug as Keytruda Rivals Loom
Big Pharma is circling Inhibrx as Keytruda's patent cliff nears, with INBRX-106 alone valued at more than $8 billion and the pipeline topping $9 billion.

Big Pharma’s hunt for the next Keytruda extender has put Inhibrx Biosciences in the crosshairs, with Merck & Co, Merck KGaA and Ono Pharmaceutical among the companies weighing an experimental cancer drug that could be worth more than $8 billion on its own. The prize is not just another oncology asset. It is a bet on whether Inhibrx can help stretch the commercial runway of Merck’s blockbuster immunotherapy before cheaper biosimilar competition eventually chips away at one of the world’s best-selling medicines.
The lead candidate is INBRX-106, an antibody designed to stimulate the immune system by activating a receptor on T cells. Inhibrx has been testing it in combination with Keytruda against Keytruda alone in first-line unresectable or metastatic head and neck squamous cell carcinoma. On December 16, 2025, the company said it had recruited 46 of the 60 patients in the randomized Phase 2 portion of the Phase 2/3 trial, giving the program a clearer path to a near-term readout and keeping deal interest alive. Early data have been strong enough to fuel expectations that response rates could improve meaningfully if the drug holds up in larger testing.
The company’s second asset, ozekibart, has added to the valuation argument. Inhibrx disclosed on April 21 that the FDA granted fast track designation in January 2021 and orphan drug designation in November 2021 for chondrosarcoma. In the same update, Inhibrx said 42% of patients remained progression-free at the six-month landmark in its colorectal cancer data, while the disease control rate reached 87%. Those results, along with the possibility of an eventual filing for approval, suggest ozekibart could become more than a side bet.
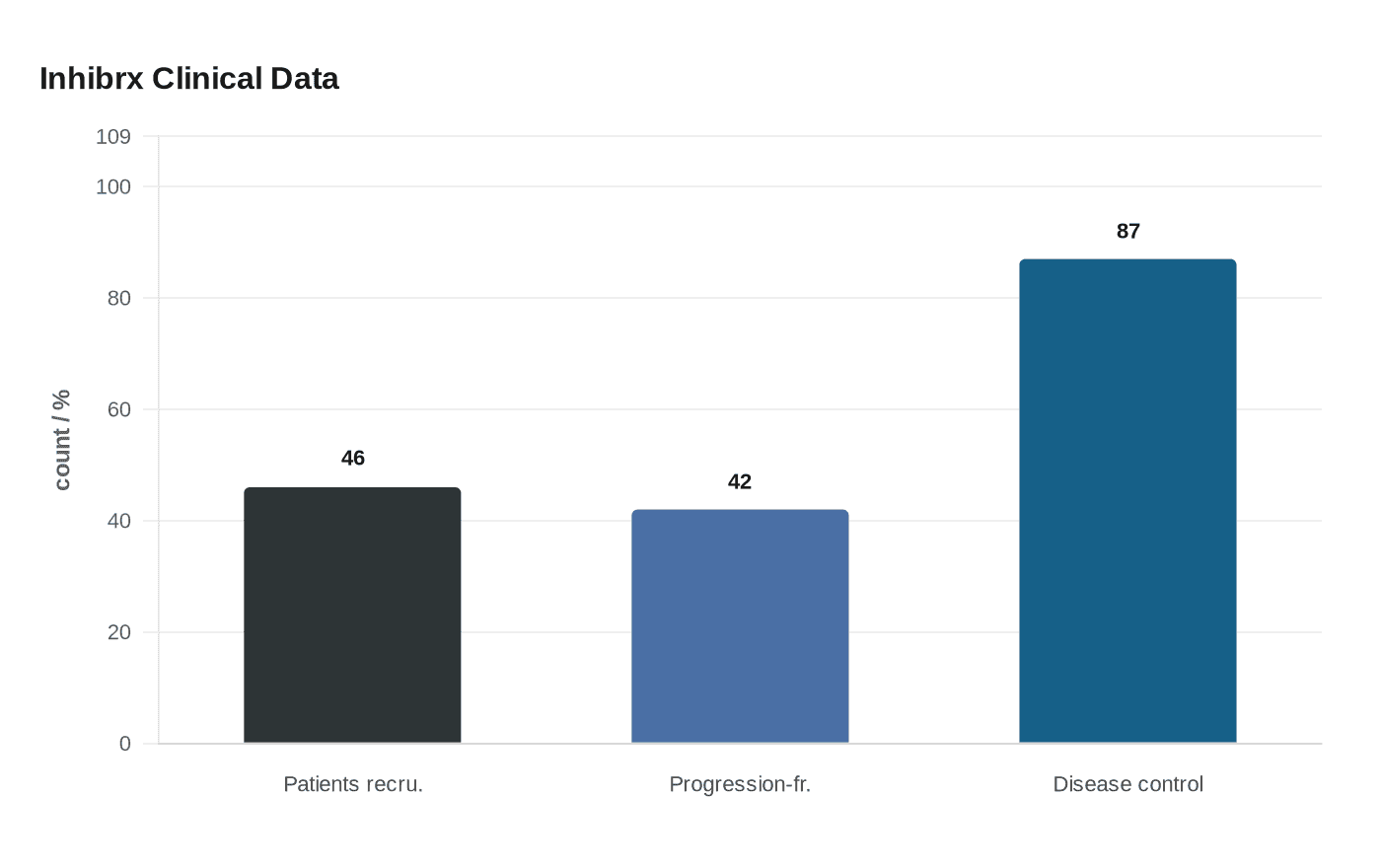
Together, the two programs could be worth more than $9 billion if clinical trials continue to meet expectations. Inhibrx shares jumped after the report, a sign of how quickly investors reprice any credible oncology deal pipeline. Any transaction still appears several months away, but the logic is familiar across drugmakers: buy growth before the market fully proves it, and pay for pipeline insurance while the patent clock is still ticking on the industry’s biggest revenue engine.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip

