Eli Lilly Drug Could Give Patients a Needle-Free Treatment Option
The FDA approved Eli Lilly's orforglipron pill, marketed as Foundayo, offering obesity patients a once-daily needle-free option starting at $25/month for insured patients.
The Food and Drug Administration approved Eli Lilly's once-daily obesity pill orforglipron on Wednesday, giving patients who want to avoid injections a new treatment option under the brand name Foundayo and setting up a competitive battle with Novo Nordisk in the rapidly growing oral GLP-1 market.
The approval could expand access to weight-loss treatment for millions of Americans who have been unable or unwilling to use injectable drugs because of cost, needle aversion, or strict dosing requirements. Obesity affects more than 40% of U.S. adults.
Eli Lilly said shipments of Foundayo are expected to begin Monday. With the company's discount card, insured patients may be able to access the drug starting at $25 per month. For patients paying cash, monthly prices will range from $149 to $349, depending on the dose.
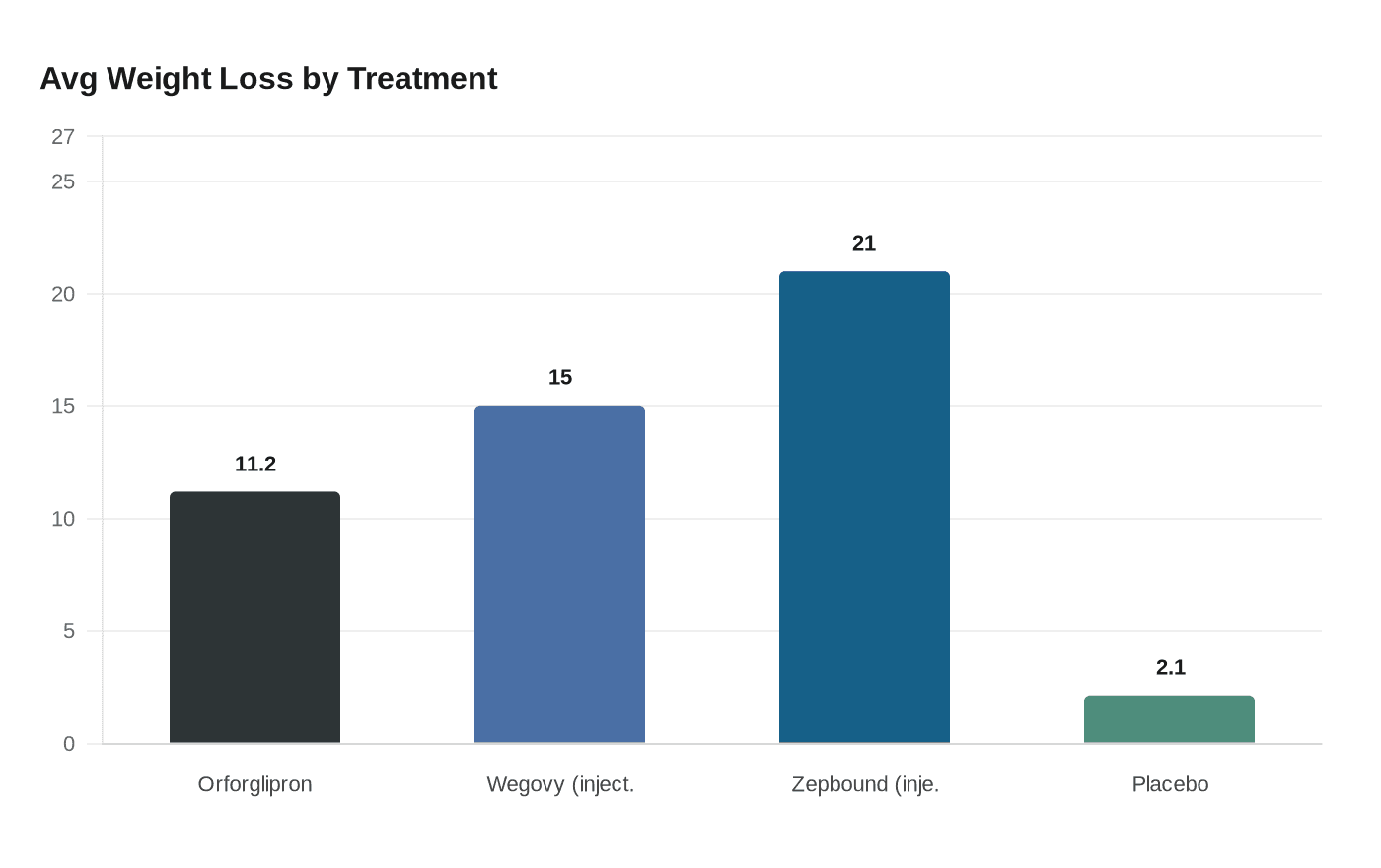
In a clinical trial involving more than 3,000 adults with obesity, participants who received the highest dose of orforglipron, 36 milligrams, lost an average of 11.2% of their body weight, or about 25 pounds, over more than 16 months. That compared with an average weight loss of 2.1%, or less than 5 pounds, among patients who received a placebo, according to results published in the New England Journal of Medicine. Participants taking the drug also saw improvements in waist circumference, blood pressure, triglycerides, and cholesterol levels.

While the daily pill offers greater convenience, it produced less weight loss than injectable GLP-1 treatments. Lilly's injectable Zepbound results in an average weight loss of about 21%, while Novo Nordisk's injectable Wegovy averages around 15%.
One of Foundayo's key advantages over competing oral options is its lack of dosing restrictions. Unlike Novo Nordisk's oral pill, which must be taken with a sip of water on an empty stomach followed by a 30-minute wait before eating or drinking, orforglipron can be taken without such restrictions.
The FDA approved the drug through a new fast-track review program aimed at shortening approval timelines, saying it reviewed Lilly's application in just 50 days.
Lilly CEO David Ricks said shots haven't been as big of a barrier to uptake as Lilly once thought they would be, but he still sees Foundayo as an attractive option for people who would rather take a pill or who are searching for a lower price than the injectables. He also views the drug as a tool for maintenance, for patients who have already reached their goal weight on an injectable and want to sustain that progress.
Because orforglipron is a small molecule rather than a peptide, it does not require cold-chain storage, unlike Zepbound and Wegovy. "[Foundayo] does allow for scalability, and that will allow us to launch this globally on the first instance," Ricks said.
Gastrointestinal side effects led between 5% and 10% of participants taking orforglipron to discontinue treatment, compared with nearly 3% of those in the placebo group.
Orforglipron joins Novo Nordisk's oral version of Wegovy, which has generated more than 600,000 prescriptions in the United States since its approval. About one in eight people in the U.S. have used injectable GLP-1 drugs, but high costs have limited access for many patients, a gap Lilly is positioning Foundayo to help close.
Know something we missed? Have a correction or additional information?
Submit a Tip

