FDA Accepts Telix Resubmitted NDA for Glioma Imaging Agent Pixclara
FDA set a Sept. 11 deadline to rule on Pixclara, a glioma PET tracer that could help U.S. oncologists separate tumor recurrence from radiation damage without biopsy.

The FDA accepted Telix Pharmaceuticals' resubmitted New Drug Application for TLX101-Px, commercially known as Pixclara, assigning the 18F-FET PET imaging agent a Prescription Drug User Fee Act goal date of September 11, 2026. The acceptance reopens a regulatory clock that now gives the agency roughly five months to render a decision on a tracer that neuro-oncologists in Europe and Australia have already folded into standard clinical workflows.
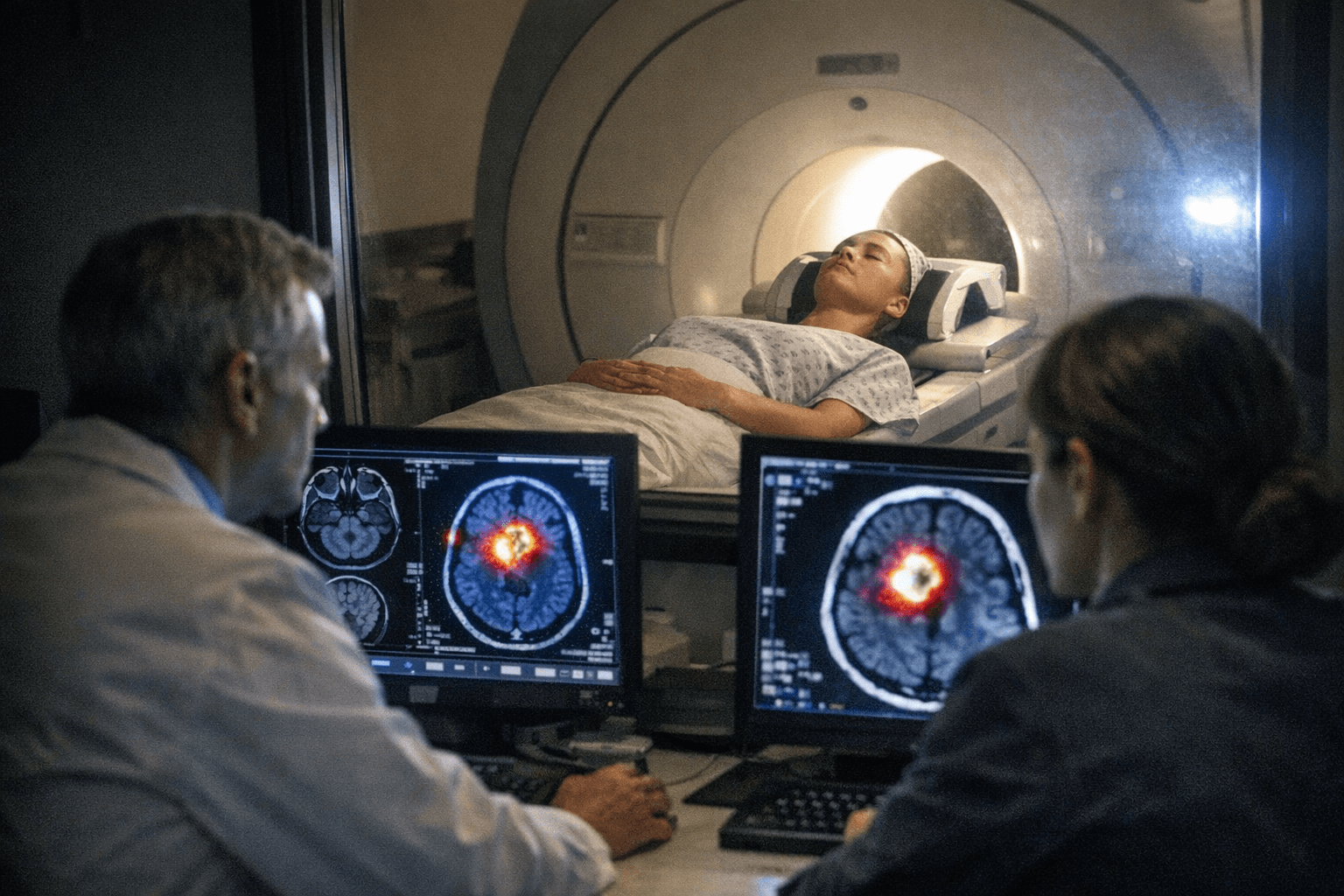
The core diagnostic problem Pixclara aims to solve is one that haunts glioma care after treatment: MRI often cannot reliably tell whether a new or growing lesion represents the tumor coming back or radiation necrosis and other therapy-related tissue changes. That ambiguity can lead to delayed treatment, unnecessary surgery, or misclassification of a patient's disease status. Thomas Hope, MD, Vice Chair of Radiology and Biomedical Imaging at UCSF, pointed to a "critical unmet need in improving our ability to image residual glioma after treatment," noting that FET-PET has already been used broadly in clinical practice internationally. Antonio Chiocca, MD, PhD, of Mass General Brigham Cancer Institute called the FDA's acceptance "a meaningful step toward broader access for patients and clinicians in the United States," framing sharper diagnostic tools as a lever for better treatment decisions.
18F-FET, or Floretyrosine F-18, is a radiolabeled amino acid tracer that cancer cells take up via the LAT1 transporter at elevated rates, producing metabolic signal that conventional contrast-enhanced MRI cannot capture. That biological mechanism is already reflected in international neuro-oncology practice guidelines, but U.S. oncologists have had no FDA-cleared version of the agent, leaving American imaging centers without a regulatory pathway to routinely offer the scan.
Telix holds both Fast Track and Orphan Drug designations for TLX101-Px, recognition of glioma's severity and its relatively limited patient population. Those designations carry procedural advantages, including more frequent FDA interactions during review and potential exclusivity incentives if the agency grants approval. The September PDUFA date sets the outer boundary of that expedited review clock.
The company also flagged that TLX101-Px's LAT1-targeting mechanism connects diagnostically to its broader therapeutic pipeline, raising the possibility of future theranostic strategies in which the same biological target guides both imaging and treatment. That longer-term vision depends first on clearing the regulatory hurdle now formally in motion.
Whether approval translates into clinical reach is a separate and harder question. PET radiopharmaceuticals require short-lived isotope production at or near imaging facilities, meaning distribution logistics constrain adoption in ways that oral or intravenous drugs do not. Imaging centers would need access to fluorine-18 production capacity and validated delivery protocols. Reimbursement coverage by Medicare and private insurers would then determine whether ordering the scan is financially viable for hospitals outside major academic neuro-oncology programs. Telix has not publicly detailed its U.S. manufacturing and distribution plan, and payer decisions on coverage typically lag FDA approval by months or longer. With the September 11 deadline now fixed, those commercial and infrastructure questions move to the foreground.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip