Hundreds Describe Steroid Withdrawal Skin Condition Doctors Refuse to Recognise
Hundreds described steroid withdrawal to the BBC as a living hell; one survey found 26% of patients still symptomatic more than five years after stopping steroids.
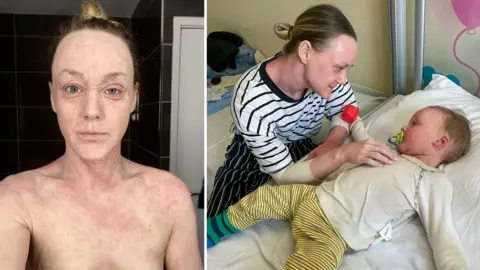
Hundreds of people have described to the BBC a skin condition they say was triggered by stopping prescription steroid creams, one they call worse than the original eczema the treatment was meant to address. Their accounts converge around a single label: Topical Steroid Withdrawal, or TSW. Most of their doctors do not believe it exists as a distinct clinical condition.
TSW, also known as Red Skin Syndrome, topical steroid addiction, or topical steroid rebound phenomena, is characterised by burning, peeling, cracking, and intensely itchy skin that patients report emerging after they discontinue topical corticosteroid creams. Symptoms have been documented after as little as two weeks of steroid use, though the majority of cases are tied to medium-to-high-potency steroids used for 12 weeks or more. Treatment duration of six months or longer appears in 63% of reported cases as a major risk factor. One survey found that 26% of patients who had stopped using topical corticosteroids for over five years were still experiencing symptoms, a figure that sits uneasily alongside the lack of formal research.
The UK's Medicines and Healthcare products Regulatory Agency formally recognised TSW reactions in September 2021, publishing guidance advising prescribers to "consider the lowest potency needed" and remain "vigilant for the signs and symptoms of topical steroid withdrawal reactions." Crucially, that guidance does not endorse abruptly stopping steroid creams; the MHRA's position implies supervised, gradual reduction rather than sudden cessation, which can itself precipitate the rebound reactions patients describe. Between the 2021 recognition and updated guidance issued in May 2024, 267 Yellow Card adverse event reports related to TSW were submitted. Yet the MHRA's 2024 update acknowledged there remained "little new information" in the scientific literature and still no accepted clinical definition.
A clinical case series published in PubMed identified 19 TSW cases, 15 of them women and 4 men, with most patients under 35 years old. Eighteen of the 19 had an underlying diagnosis of atopic dermatitis, the very condition the steroids were originally prescribed to treat. That overlap means TSW symptoms are routinely misread as an eczema flare-up, and it may explain why the MHRA found patients struggling to obtain a diagnosis. Many, dismissed repeatedly by dermatologists, have migrated to unregulated online communities, some of which promote unvetted treatments. A 2025 review in JAAD Reviews described TSW as "a rare condition associated with long-term, inappropriate use of high-potency TCSs, mainly on the face," a characterisation that patient groups say understates both the severity and the breadth of the condition they experience.

In May 2024, the MHRA mandated new potency labels on all topical steroid products sold in the UK, requiring packaging to carry the designations "mild", "moderate", "strong", or "very strong." Andrew Proctor, chief executive of the National Eczema Society, welcomed the decision.
The biological mechanisms behind TSW are also beginning to be investigated. A pilot study from researchers at the National Institutes of Health offered preliminary evidence that TSW may be driven by an over-activation of mitochondrial complex I, generating excess NAD+ and driving tryptophan toward neurotoxic metabolites including kynurenic acid. Potential treatments under investigation include the complex I blockers berberine and metformin, as well as the JAK inhibitor ruxolitinib cream. The British Journal of Dermatology published research in 2025 on developing formal TSW diagnostic criteria through a Modified Delphi Method, the most concrete step toward clinical consensus since Australian GP Dr Belinda Sheary began publishing on the condition in 2016.
Until those criteria are formalised and treatment trials move beyond pilot stage, patients using any topical steroid for longer than three months face a system with no reliable pathway: recognised by the regulator, absent from diagnostic manuals, and largely invisible to the specialists they are sent to see.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip

