FDA approves Bizengri for rare bile duct cancer, using fast-track voucher program
The FDA cleared Bizengri for a tiny biomarker-defined group of bile duct cancer patients, after a review meant to take weeks instead of months.

The Food and Drug Administration has expanded Bizengri into one of oncology’s narrowest patient groups: adults with advanced cholangiocarcinoma, or bile duct cancer, whose tumors carry an NRG1 gene fusion and whose disease has progressed after earlier systemic therapy. The decision matters because this is not a broad bile duct cancer drug. It is aimed at a rare molecular subset, the kind oncologists now look for when standard treatment has already failed.
Bizengri, also known as zenocutuzumab-zbco, had already won FDA approval on December 4, 2024, for adults with NRG1 fusion-positive non-small cell lung cancer and pancreatic adenocarcinoma. FDA materials describe it as a HER2- and HER3-directed bispecific antibody. The new cholangiocarcinoma clearance extends that label into another hard-to-treat cancer, where late diagnosis and limited treatment options often leave patients with little room to maneuver.

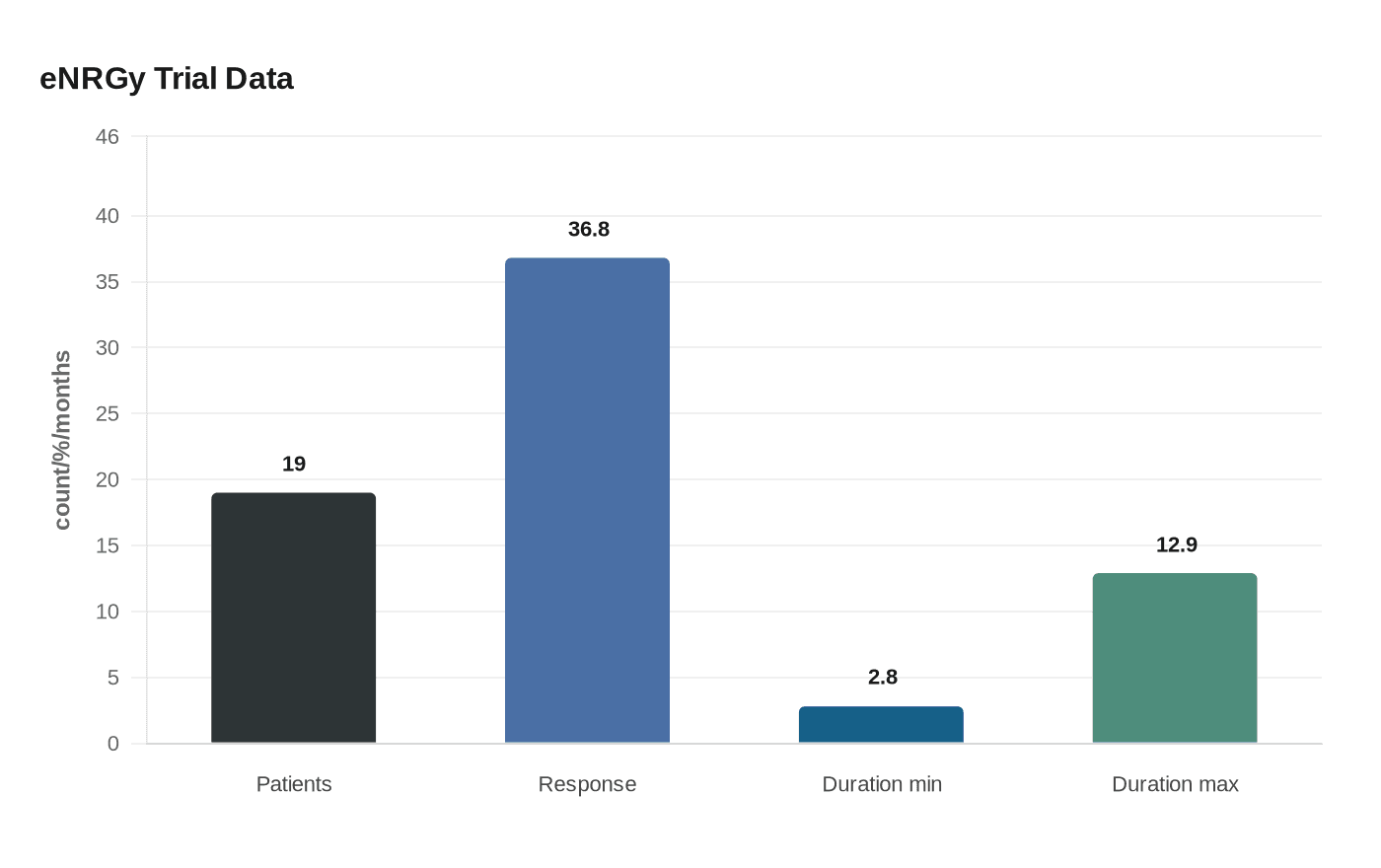
The approval was based on the phase 2 eNRGy trial, specifically the cholangiocarcinoma cohort. In the FDA’s cited data set, 19 patients were evaluable and 36.8% saw their tumors shrink. Those responses lasted from 2.8 months to 12.9 months. That level of activity is meaningful for a rare cancer with few targeted options, but it is also a reminder that the benefit was seen in a minority of patients, not across the full group.
That mixed picture is central to how families and physicians should read the decision. The drug appears capable of producing real tumor shrinkage in some patients with the right mutation, yet it is not a universal solution. The FDA also listed risks that include infusion reactions, lung inflammation and heart-related problems, along with more common side effects such as diarrhea, fatigue, nausea, shortness of breath and rashes. Bizengri’s earlier label also carries a boxed warning for embryo-fetal toxicity.
The pace of the review adds another layer to the story. Partner Therapeutics said the FDA awarded Bizengri a Commissioner’s National Priority Voucher on May 6, 2026, and the agency announced approval two days later. The pilot program, launched in June 2025, is designed to cut review time from roughly 10 to 12 months to about one to two months through a more collaborative process. For patients with advanced cholangiocarcinoma, that compressed timeline can translate into faster access to a treatment that may matter most after other options have run out.
Know something we missed? Have a correction or additional information?
Submit a Tip