FDA approves Bizengri for rare bile duct cancer with NRG1 fusion
The FDA cleared Bizengri for a tiny slice of bile duct cancer patients, but only those with an NRG1 fusion found through genetic testing.

A new FDA approval gave patients with advanced cholangiocarcinoma a drug aimed at one of the cancer’s rarest genetic drivers, but the treatment will only reach the small minority whose tumors carry an NRG1 fusion.
The agency approved Bizengri, also known as zenocutuzumab-zbco, on May 8 for adults with advanced, unresectable or metastatic cholangiocarcinoma after progression on prior systemic therapy, provided their tumors harbor an NRG1 gene fusion. It is the first approved therapy for this specific bile duct cancer subtype, a milestone in a disease that is often discovered too late for surgery and has few good options once it spreads.
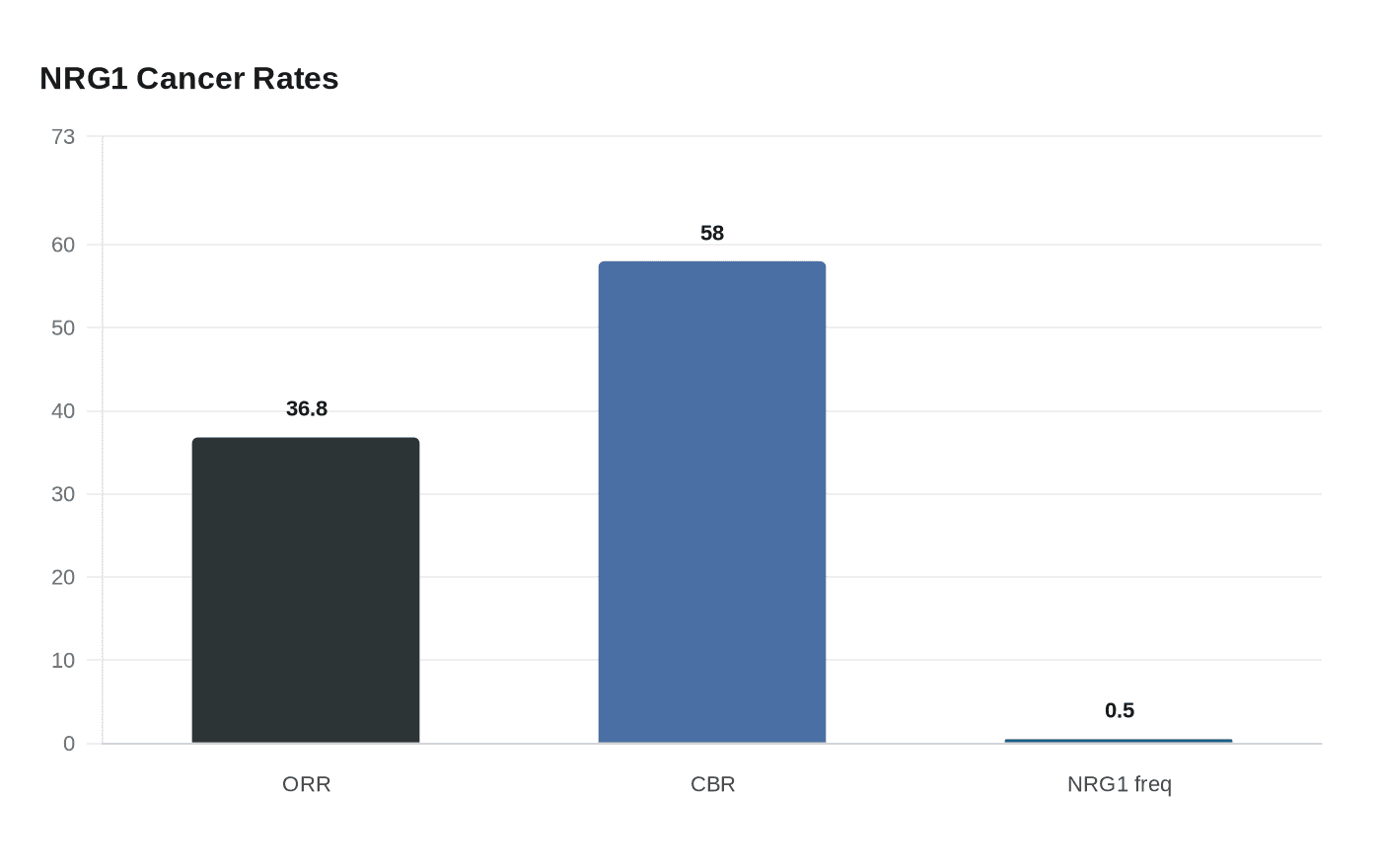
Bizengri’s case rested on the phase 2 eNRGy trial, an open-label, multicenter study of solid tumors with NRG1 fusions. Among 19 efficacy-evaluable cholangiocarcinoma patients, the objective response rate was 36.8 percent to 37 percent. The median duration of response was 7.4 months, median time to response was 1.9 months and median progression-free survival was 9.2 months. The clinical benefit rate was 58 percent, and the median age of enrolled cholangiocarcinoma patients was 57. By April 2025, 22 patients with NRG1-positive cholangiocarcinoma had been enrolled.
That narrow response is also the point: NRG1 fusions are estimated to appear in about 0.5 percent of cholangiocarcinomas. For most patients, the real barrier is not getting the drug but getting identified as eligible in the first place. The approval makes NRG1 fusion testing the gatekeeper, and how widely oncologists can order that testing, and how quickly insurers will pay for it, will help determine how much this drug changes care beyond a small molecular subset.
The stakes are high because cholangiocarcinoma remains a lethal diagnosis. CDC reporting cited death rates of 1.66 per 100,000 persons for intrahepatic bile duct cancer and 0.45 per 100,000 for other biliary tract cancers. Bizengri had already received Breakthrough Therapy and Orphan Drug designations, and the new approval expands its reach beyond the lung and pancreatic cancers it was cleared to treat in 2024.
FDA Commissioner Marty Makary said, “Patients with this ultra-rare type of cancer desperately need new treatment options.” The approval was the seventh under the FDA’s National Priority Voucher pilot program, a fast-track effort aimed at serious unmet medical needs. Even so, Bizengri carries meaningful risks, including infusion reactions, lung inflammation and heart-related side effects, so access to testing, coverage and close medical monitoring will shape how much benefit this precision-medicine advance can deliver.
Know something we missed? Have a correction or additional information?
Submit a Tip

