FDA Approves Endospan NEXUS Stent Graft System for Complex Aortic Arch Disease
The FDA cleared Endospan's NEXUS stent graft system for aortic arch repair, giving high-risk patients a minimally invasive option backed by the TRIOMPHE IDE study.
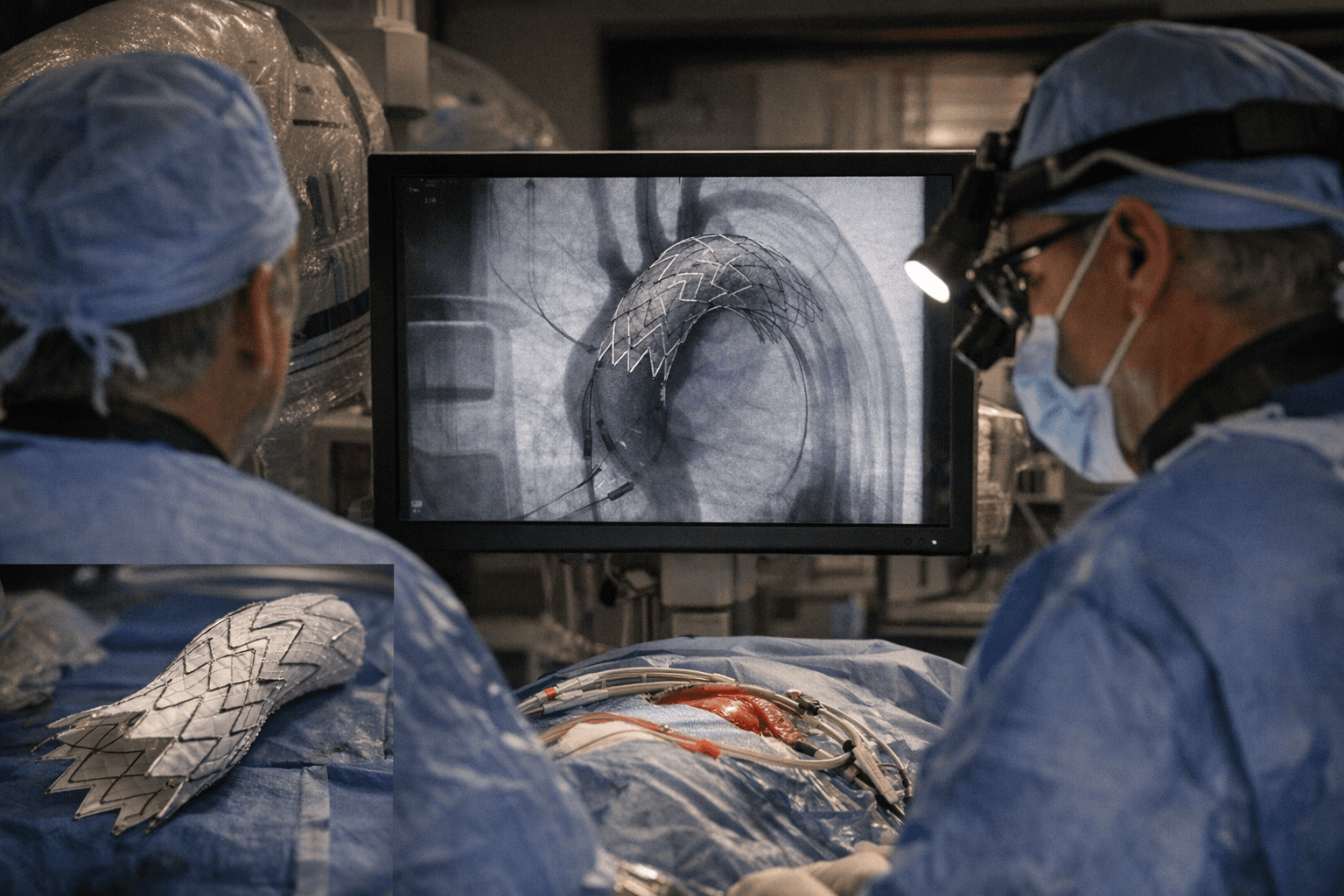
The FDA cleared Endospan's NEXUS Aortic Arch Stent Graft System for commercial use in the United States, opening an endovascular treatment path for patients with complex aortic arch disease who face prohibitive risk from conventional open surgery.
The agency's decision, supported by one-year data from the TRIOMPHE Investigational Device Exemption study, advances the first FDA-approved endovascular option targeting the ascending aorta and arch, a segment historically resistant to transcatheter approaches. The TRIOMPHE trial, a prospective multicenter study, demonstrated the system could safely and effectively treat chronic aortic dissections and other arch pathologies in high-risk surgical candidates.
Engineered as a bimodular system, the NEXUS device incorporates a low-profile 20F delivery system and a pre-shaped catheter designed to achieve a single pass into the arch, reducing the manipulation typically required in that anatomical region. An integrated branch is built into the system to optimize hemodynamics, with the overall design intended to mimic the natural geometry of the ascending aorta and arch.
Brad Leshnower, national cardiac surgery co-principal investigator on the TRIOMPHE study, and Dr. Ross Milner, Louis Block Professor of Surgery and chief of vascular surgery and endovascular therapy, both acknowledged the investigational team's contributions and pointed to the significance of a minimally invasive option for patients whose life-threatening aortic disease places open repair beyond reach.
The clinical need is substantial. Endospan estimates more than 120,000 patients suffer thoracic aortic arch disease annually across the United States and Europe, yet only about a quarter of those patients are ever diagnosed or treated. For individuals whose anatomy or comorbidities rule out open surgery, the NEXUS approval creates a meaningful alternative where few existed.
The U.S. commercial launch will face practical hurdles including physician training, reimbursement pathways, and device availability. Broader adoption will also depend on post-approval outcomes as real-world data accumulates beyond the TRIOMPHE trial's one-year window. In a cardiovascular device market that has seen limited endovascular innovation for ascending aortic pathologies, that is a high-stakes test Endospan will now have to pass outside the controlled setting of a clinical trial.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip