FDA Approves First Cerebral Venous Stent for Severe Intracranial Hypertension
The FDA cleared Serenity Medical's River stent for IIH patients who've failed six months of treatment, the first purpose-built cerebral venous device to win U.S. approval.
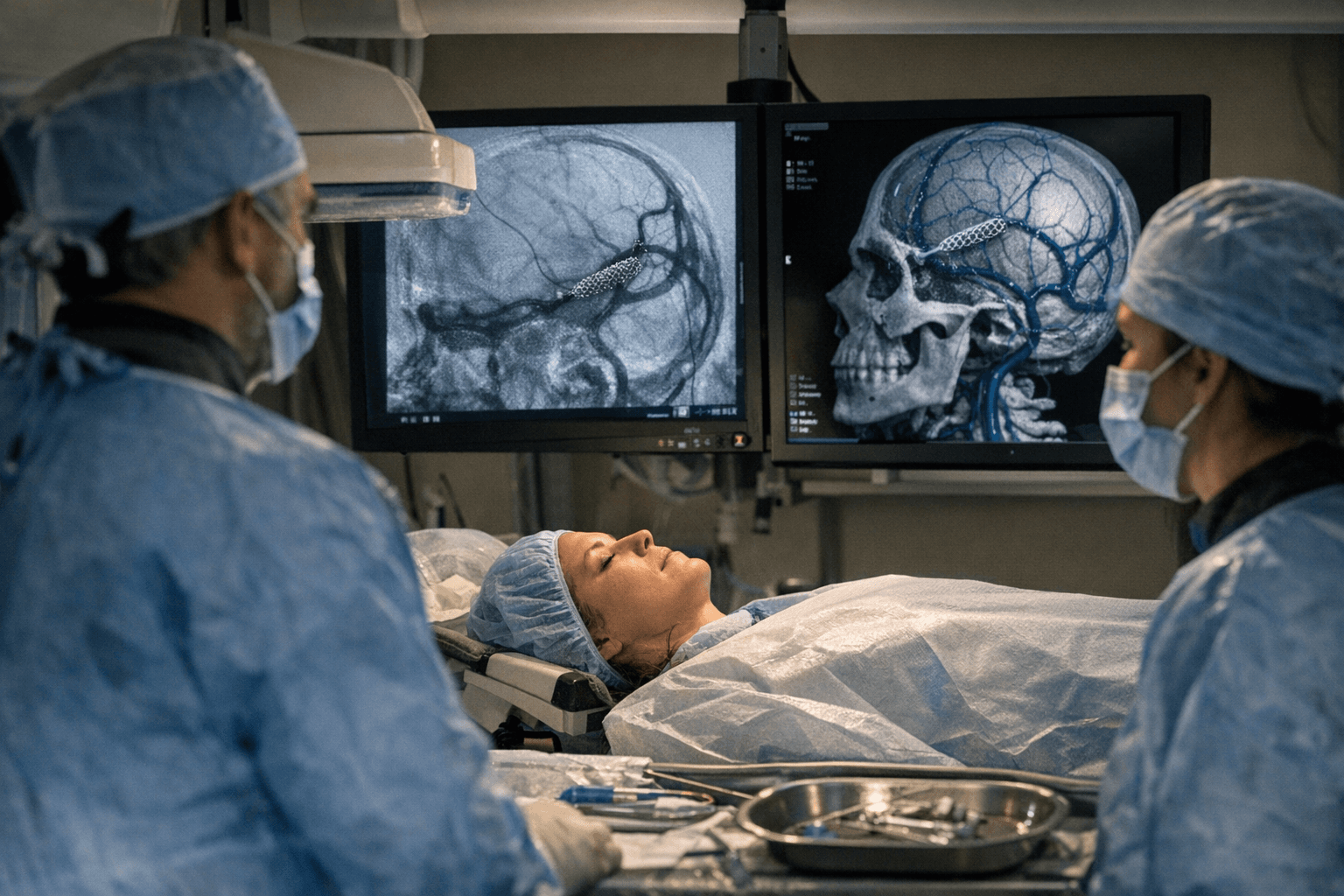
For the roughly 200,000 Americans living with idiopathic intracranial hypertension, the condition has long existed in a medical paradox: serious enough to steal vision and dominate daily life, yet rare enough that the device industry largely left it without a purpose-built tool. The FDA's clearance of Serenity Medical's River stent, granted March 31 through the Humanitarian Device Exemption pathway, begins to close that gap.
The HDE is a regulatory pathway established by Congress specifically for medical devices targeting diseases or conditions that affect fewer than 8,000 individuals per year in the United States. Unlike the standard premarket approval process, it does not require manufacturers to demonstrate full effectiveness; instead, they must show the device is safe and offers probable benefit. That lower evidentiary bar is not a loophole so much as a deliberate policy accommodation: for rare conditions, assembling the tens of thousands of patients needed for a large randomized controlled trial is often simply not feasible.
After HDE approval, however, manufacturers face meaningful post-market obligations. The device must be administered only in facilities with institutional review board oversight, and IRB or appropriate local committee approval is required before the device can be used at any facility for clinical care. That structure means the River stent's rollout will not function like a typical commercial product launch. Serenity Medical's commercialization partner, Radical Technologies, will work with specialized neurovascular centers to establish training, procedural protocols and facility-level oversight before the stent reaches patients.
The underlying clinical evidence rested on a 39-patient trial, modest by conventional standards but substantial given the disease population. Principal investigator Athos Patsalides, of North Shore University Hospital in Manhasset, New York, and colleagues described the River stent as "the first stent specifically designed for intracranial venous sinuses," distinguishing it from the off-label arterial stents clinicians had been adapting for years. The prospective, open-label, multi-center, single-arm River Study enrolled subjects across five U.S. centers; its one-year results were published in the Journal of Neurointerventional Surgery in February 2025.
Venous sinus stenting procedures for IIH have been rising sharply, increasing approximately 80 percent per year, with a parallel yearly decrease in ventriculoperitoneal shunting procedures of roughly 54 percent. The acceleration reflects growing clinical recognition that venous sinus stenosis is a key driver of elevated intracranial pressure in many IIH patients, but it has also outpaced regulatory clarity. Until now, every procedure was performed with a device not cleared for that specific use.
Who actually qualifies for the River stent is defined narrowly. The FDA approved its humanitarian use for adult patients with IIH and significant venous sinus stenosis who are resistant or intolerant to medications. It is reserved for patients with severe headaches who have failed to respond to six months of medical therapy, including attempts at weight loss, or for patients who have had visual symptoms or signs that are vision-threatening despite medical therapy.
The insurance landscape remains unsettled. Aetna's existing clinical policy considers venous stent placement in the transverse sinus medically necessary for refractory IIH when specific ophthalmologic, neurologic, neuroimaging and intracranial pressure criteria are met. Medicare had previously classified venous sinus stenting as investigational; the FDA's HDE approval may prompt payers to revisit those determinations, though coverage decisions rarely follow regulatory clearance automatically.
IIH disproportionately affects women between the ages of 20 and 50 and is strongly associated with obesity, a demographic with well-documented barriers to specialist access and procedural care. The condition is characterized by elevated intracranial pressure, severe headaches, progressive vision loss and cognitive difficulties, often in the absence of any tumor or structural abnormality.
Y. Pierre Gobin, founder of Serenity Medical and a neurointerventional specialist at Weill-Cornell Medicine, captured both the clinical and human dimensions of the approval: "Delivering the first venous stent approved for severe, refractory IIH is incredibly meaningful to our dedicated team and to people living with IIH who have had limited options for relief until now."
For clinicians, the clearance shifts the conversation from whether to use a venous stent to how to select the right patients and track outcomes rigorously. Post-approval registries will be central to that effort, serving as the evidence infrastructure that a 39-subject trial, however carefully conducted, cannot fully provide. The question of long-term durability, restenosis rates and how the River stent compares to shunting procedures in vision preservation will be answered in the years after implantation, not before.
Know something we missed? Have a correction or additional information?
Submit a Tip

