FDA Approves High-Dose SPINRAZA Regimen, Expanding Options for SMA Patients
A stronger SPINRAZA regimen just cleared the FDA, with DEVOTE trial data showing significant motor gains in infants but a first-year price tag near $750,000.
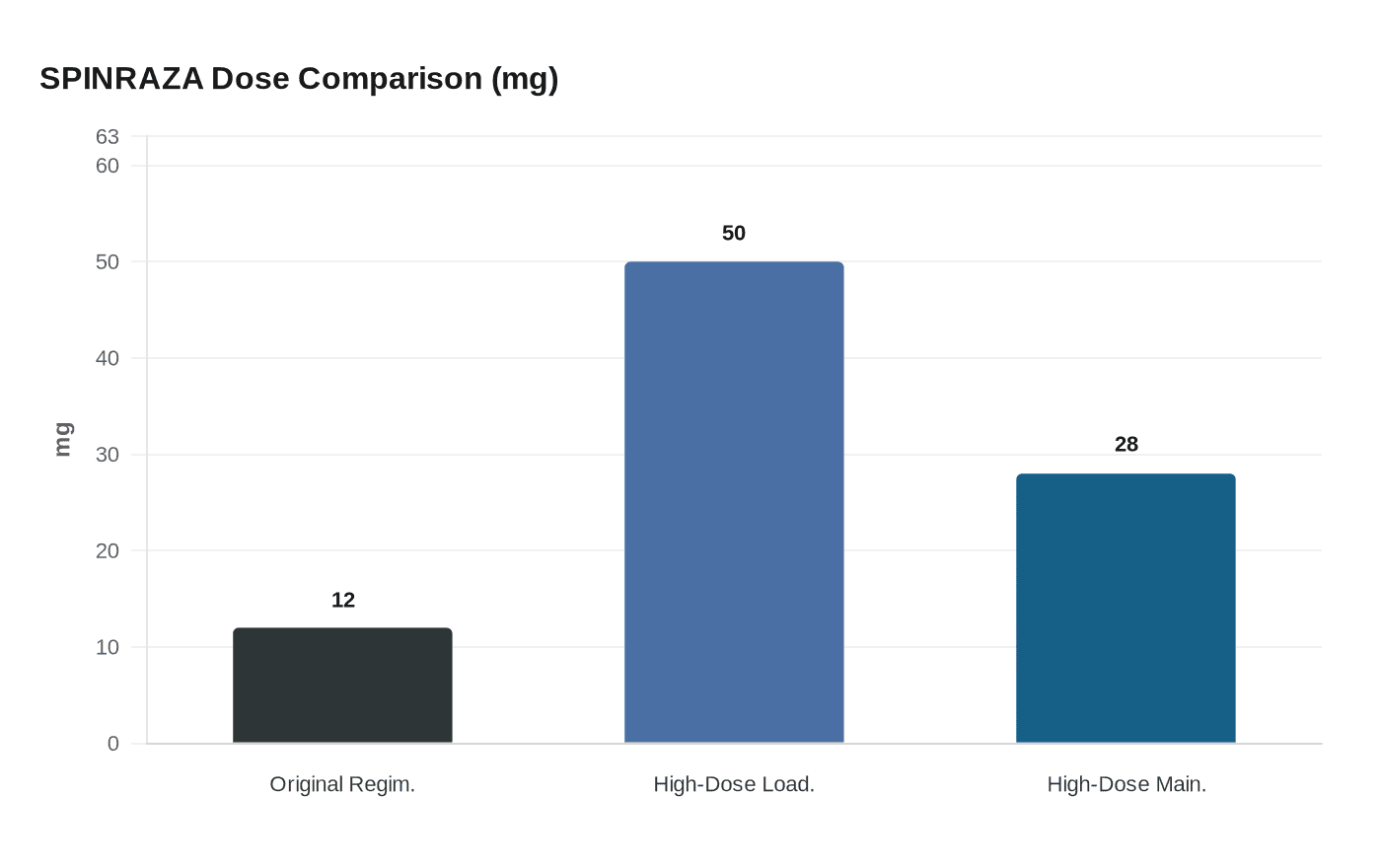
The FDA approved a higher-dose regimen of SPINRAZA (nusinersen) for spinal muscular atrophy on March 30, marking the first major labeling change to the pioneering antisense therapy since its initial authorization a decade ago. The new schedule, comprising 50 mg/5 mL loading doses and 28 mg/5 mL maintenance doses, roughly triples the concentration delivered per injection compared to the original 12 mg regimen and is approved for both newly diagnosed patients and those already receiving the standard dose.
The regulatory decision rested on data from the three-part, Phase 2/3 DEVOTE study, which enrolled 145 participants across ages and SMA types at approximately 42 clinical sites globally. In the pivotal Part B cohort, 75 treatment-naïve children with infantile-onset SMA were randomized to either the high-dose regimen or the approved 12 mg schedule. Treated infants experienced statistically significant improvements in motor function, as measured by the Children's Hospital of Philadelphia Infant Test of Neuromuscular Disorders at six months. The high-dose arm also produced greater reductions in neurofilament light chain, a biomarker of neurodegeneration, and improved event-free survival.
"The FDA approval of the High Dose regimen of SPINRAZA is a major advancement for our community," said Kenneth Hobby, president of Cure SMA. Richard Finkel, director of the Center for Experimental Neurotherapeutics at St. Jude Children's Research Hospital, said the regimen "demonstrated meaningful clinical benefit while maintaining a well characterized safety profile." Cure SMA's chief scientific officer, Dr. Jackie Glascock, said the organization would work with Biogen and providers to ensure patients have a clear path to the new option.
That path carries real financial weight. SPINRAZA currently carries a list price of roughly $125,000 per vial, translating to approximately $750,000 in year one and around $375,000 annually after that. Every major insurer requires prior authorization, typically demanding genetic confirmation of 5q SMA, a neurologist's prescription, and documented baseline motor function assessments before coverage proceeds. Because SPINRAZA is delivered as an intrathecal bolus through lumbar puncture, each dose requires a hospital or infusion-center visit by a specialist experienced in spinal procedures. Patients with spinal fusions, a complication that becomes more common as SMA progresses, may face additional complexity accessing the intrathecal space and may need interventional radiology support.
For patients already on the standard regimen, DEVOTE's open-label cohort showed that transitioning to the high dose was feasible and associated with meaningful functional improvement on the Hammersmith Functional Motor Scale. That finding puts the upgrade squarely on the table for current patients and their neurologists. Biogen said high-dose SPINRAZA will be available in the United States within weeks; it is already approved in the European Union, Switzerland, and Japan.
The approval also reconfigures competitive dynamics in an SMA field anchored by three treatment modalities: SPINRAZA's repeated intrathecal injections, Roche's oral daily modifier Evrysdi (risdiplam), and Novartis' one-time gene-replacement therapy Zolgensma. A demonstrably more potent SPINRAZA regimen gives neurologists fresh clinical justification for the intrathecal route and gives Biogen a sharper commercial counter to therapies that had begun to narrow its market position.
Cure SMA plans to host an informational webinar on April 30 to walk patients, caregivers, and providers through transition planning and insurance coverage strategies for the new regimen.
Know something we missed? Have a correction or additional information?
Submit a Tip

