FDA Clears Medtronic OmniaSecure Lead for Left Bundle Branch Pacing
Medtronic's OmniaSecure is now the first defibrillation lead FDA-approved for the left bundle branch, with clinical trial data showing 100% defibrillation success at implant.

Medtronic's OmniaSecure defibrillation lead became the first defibrillation lead approved by the FDA for placement in the left bundle branch area when the agency granted the company an expanded indication on March 23, 2026. The distinction matters clinically: now approved for placement in the left bundle branch (LBB) area, the lead can be used for conduction system pacing (CSP), which closely mimics the heart's natural physiology.
The OmniaSecure defibrillation lead connects to an implantable cardioverter-defibrillator (ICD) or cardiac resynchronization therapy defibrillator (CRT-D) to treat potentially life-threatening ventricular tachyarrhythmias, ventricular fibrillation (VT/VF), and bradyarrhythmias. The expanded indication also opens a second therapeutic avenue: patients in need of cardiac resynchronization may benefit from left bundle branch optimized cardiac resynchronization therapy (LOT-CRT), a novel therapy that combines CSP with left-ventricular pacing to further improve patient outcomes.
The approval rests on a robust clinical foundation. FDA approval for the OmniaSecure defibrillation lead was supported by data from the global LEADR LBBAP trial, which demonstrated the lead's safety and effectiveness. Late-breaking data from the study presented at Heart Rhythm 2025, and later published in Heart Rhythm, showed that the lead delivered high defibrillation success at implant (100%) and a low OmniaSecure-related major complication rate at three months (2.1%) when placed in the LBB area for physiologic pacing. Enrollment in the LEADR LBBAP study was completed with 323 subjects enrolled. Longer-term outcomes from the study are set to be presented at HRS 2026.
"The robust body of clinical evidence from the LEADR LBBAP study supports the OmniaSecure defibrillation lead as a safe and effective way to activate physiologic pacing through the left bundle branch area or in applications like LOT-CRT, while also demonstrating reliable defibrillation success," said Pugazhendhi Vijayaraman, M.D., cardiac electrophysiologist at Geisinger Wyoming Valley Medical Center in Wilkes-Barre, Pa., and principal investigator of the LEADR LBBAP study.
"Conduction system pacing is a rapidly growing therapy for patients who need a pacemaker. Now, patients who require a defibrillator and pacing have an option that can safely deliver life-saving defibrillation therapy and activate the heart's natural electrical system to enable a more synchronous, physiologic pattern," said Trevor Cook, vice president and general manager of the Defibrillation Solutions business, which is part of the Cardiac Rhythm Management operating unit at Medtronic.
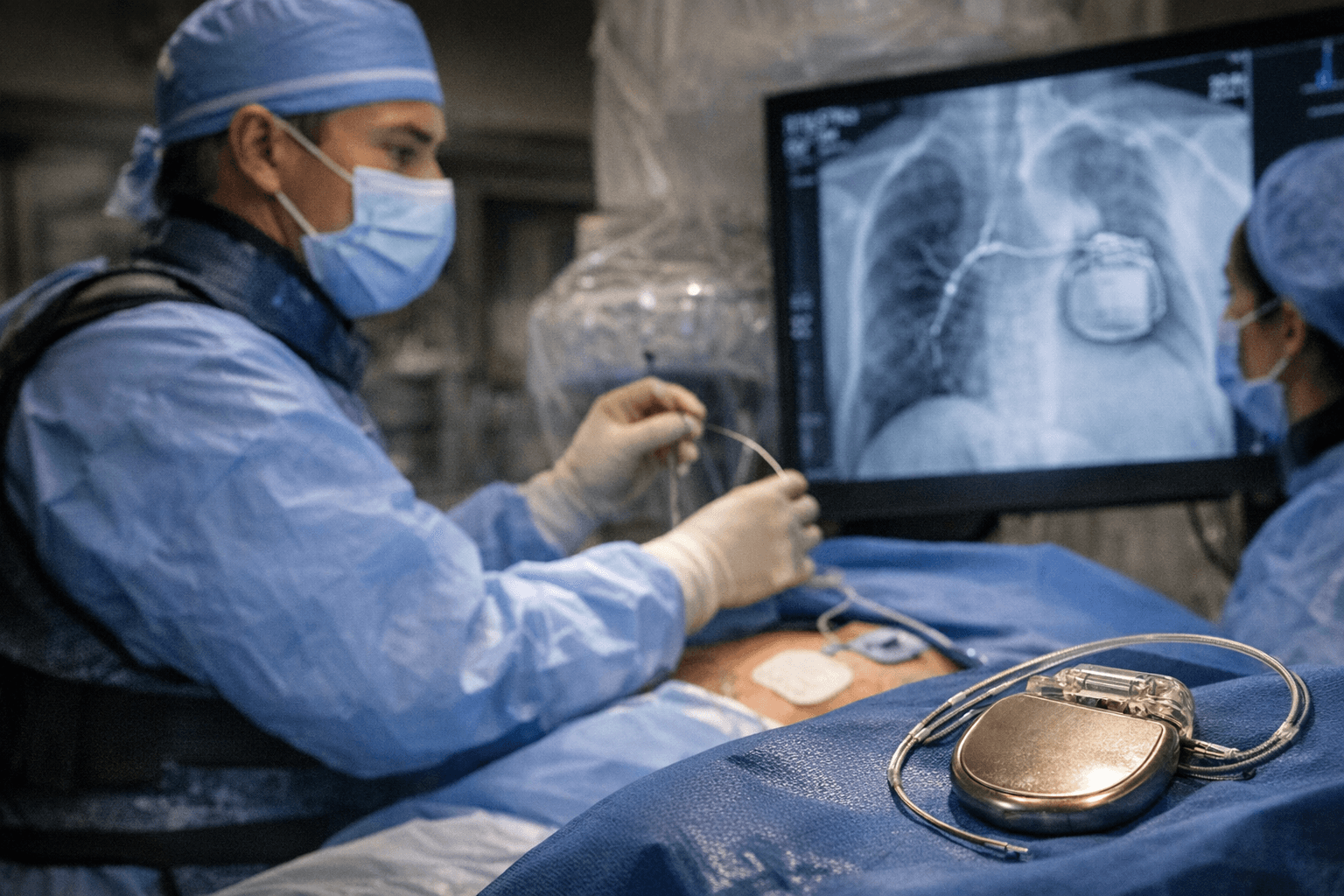
In addition to being the first defibrillation lead approved for placement in the LBB area, the OmniaSecure lead is the smallest diameter defibrillation lead on the market at 4.7 French, or 1.66 mm, and the only one approved for adults and adolescent pediatric patients ages 12 and up. MassDevice noted the scale of that measurement: the 4.7 Fr diameter equals roughly the width of the graphite in a wooden pencil.
Engineered based on the reliable Medtronic SelectSecure Model 3830 pacing lead, the first pacing lead to receive FDA approval for CSP in 2022, the OmniaSecure defibrillation lead is built for high reliability and durability with its lumenless construction, which enables the lead to be delivered via catheter for precise placement in the right ventricle, in addition to the LBB area.
The LBB-area approval is the device's second U.S. regulatory milestone in under a year. Following FDA approval for placement in traditional locations in the right ventricle, the OmniaSecure defibrillation lead launched commercially in the U.S. in January 2026. The approval expands the comprehensive Medtronic portfolio of lumenless leads and accessories approved by FDA that enable conduction system pacing. This includes the SelectSecure Model 3830 pacing lead, now implanted in more than one million patients globally, as well as the recently cleared C320LBB delivery catheter and the 5944RL rotatable connector.
OmniaSecure's expanded approval builds on Medtronic's legacy within the cardiac rhythm management devices market, which currently spans external and implantable defibrillators like the Cobalt and Crome device families, as well as pacemakers and ICD leads. The Sudden Cardiac Arrest Foundation currently predicts that 350,000 Americans experience out-of-hospital cardiac arrest per year, a patient population for which broader and more physiologic defibrillation options carry direct clinical weight.
Know something we missed? Have a correction or additional information?
Submit a Tip

