FDA expands Boehringer’s Hernexeos to first-line HER2‑mutant lung cancer
The FDA cleared HERNEXEOS for first-line use in HER2‑mutant advanced NSCLC after a 76% response rate in a 72‑patient trial, accelerating review under the CNPV program.
The U.S. Food and Drug Administration expanded accelerated approval of Boehringer Ingelheim’s oral tyrosine kinase inhibitor HERNEXEOS (zongertinib) to first-line treatment for adults with unresectable or metastatic non-squamous non‑small cell lung cancer whose tumors harbor HER2 (ERBB2) tyrosine kinase domain activating mutations, the agency and the company said late February 2026. The decision makes a once‑daily targeted oral option available to treatment‑naïve patients after a single‑arm cohort showed a high response rate.
Boehringer said the Beamion LUNG‑1 Phase Ib treatment‑naïve cohort of 72 patients produced an objective response rate of 76%, including complete responses in 11% and partial responses in 65%. The company reported that 64% of responders maintained their response for six months or longer; a separate report cited a median duration of response of 14.1 months. Boehringer framed the approval as based on objective response rate and duration of response, and warned that continued approval may be contingent on verification of clinical benefit in a confirmatory trial.
The company is already enrolling patients in Beamion LUNG‑2, a Phase III confirmatory trial listed on ClinicalTrials.gov as NCT06151574. Dosing approved by the FDA is weight based: 120 mg orally once daily for patients weighing less than 90 kg and 180 mg once daily for those 90 kg or more, taken with or without food and continued until disease progression or unacceptable toxicity, regulators said.
The review was handled through the FDA Commissioner’s National Priority Voucher pilot program, which shortens review timelines. Boehringer filed the first‑line application on Jan. 13, 2026, the company said, and FDA Commissioner Marty Makary noted, “The decision on Hernexeos came 44 days after the filing date.” The faster timeline allowed the agency to move well ahead of a typical 10 to 12 month review clock for complex oncology filings.
Boehringer executives stressed the commercial and patient impact. Itziar Canamasas, the company’s global head of oncology, said the first‑line approval “would allow Boehringer to target the entire patient population, potentially doubling its addressable market,” and that earlier use of the drug “could offer patients a more convenient, tolerable and efficacious treatment option.” Boehringer also highlighted the company statement that “HERNEXEOS® (zongertinib tablets) [was] approved based on an objective response rate of 76% (N=72) as demonstrated in the Beamion LUNG‑1 clinical trial.”
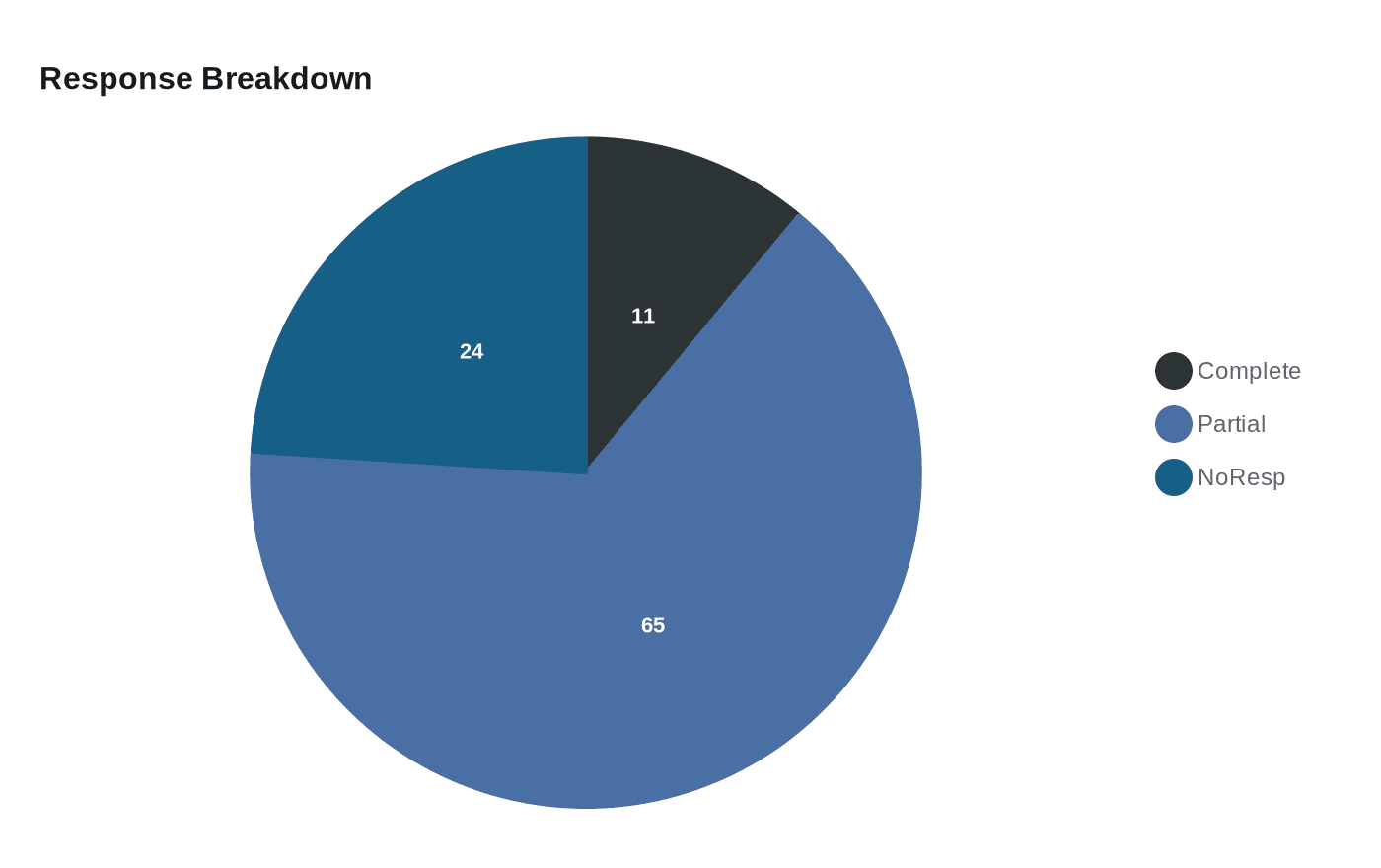
The expanded indication follows an initial accelerated approval in August 2025 for patients who had received prior systemic therapy. The move puts HERNEXEOS directly into competition with other investigational and recently approved HER2‑directed agents; Bayer’s sevabertinib was approved for second‑line use in November 2025 and has reported first‑line data but had not filed for that indication.
HER2 tyrosine kinase domain mutations are uncommon, occurring in roughly 2% to 5% of NSCLC cases depending on the estimate; some sources place the range at 2% to 4% and others at 3% to 5%. Patients with HER2‑mutant disease are more likely to be younger, female and never‑smokers and face a higher risk of brain metastases, clinical observers note.
Regulatory caveats remain: the approval is accelerated and contingent on confirmatory Phase III results. Boehringer expects readouts from its larger trials later in the year that will determine whether the FDA converts the accelerated clearance to full approval.
Know something we missed? Have a correction or additional information?
Submit a Tip

