GE HealthCare Wins FDA Clearance for Photonova Spectra Photon-Counting CT System
GE HealthCare's Photonova Spectra scored FDA 510(k) clearance, putting its Deep Silicon photon-counting CT directly against Siemens Healthineers in the U.S. market for the first time.
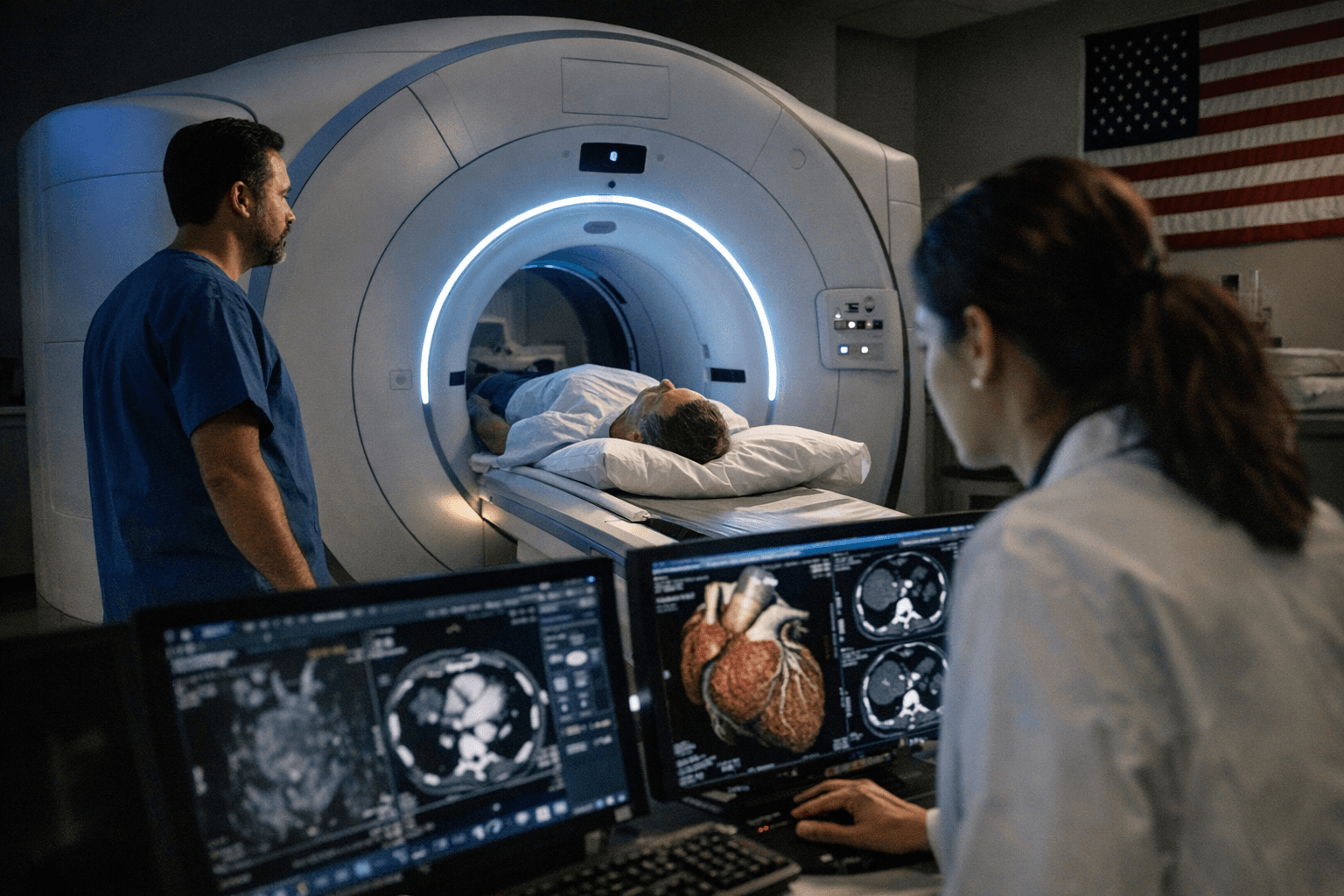
The FDA clearance positions GE HealthCare to compete with Siemens Healthineers' Naeotom Alpha, the first photon-counting CT machine to reach the U.S. market — and GE is entering that contest with technology it argues is fundamentally different from anything currently on the market.
GE HealthCare (Nasdaq: GEHC) received 510(k) clearance from the U.S. Food and Drug Administration for Photonova Spectra, an innovative photon-counting computed tomography solution powered by the company's novel Deep Silicon detector technology and offered as a flexible platform with multiple configurations to meet diverse clinical needs. The company announced the clearance from Chicago on March 23, 2026.
Photonova is the first photon-counting CT technology to use "deep silicon," a material that GE HealthCare acquired from Prismatic Sensors AB, a Swedish startup it purchased in 2020. Photonova Spectra introduces the next generation of spectral photon-counting CT technology with 8-bin energy resolution and a one-scan universal workflow designed to boost efficiency and reduce complexity across a wide range of CT exams.
The underlying physics distinguish the system from conventional scanners. Photon-counting CT represents a significant advancement in medical imaging. Unlike conventional CT systems, which first convert X-ray photons into visible light before measuring them, photon-counting CT directly counts individual photons and measures their energy. This approach enables higher spectral and spatial resolution as well as improved tissue characterization, helping provide clinicians with rich information to detect and diagnose disease. The Deep Silicon detector with 8-bin energy resolution supports advanced material separation and characterization capabilities, enabling Photonova Spectra to clearly distinguish between different materials such as iodine, calcium and fat with remarkable precision.
Its wide detector coverage and rapid rotation speed of 0.23 seconds also support fast acquisition and motion-free imaging, even in challenging patient scenarios. Photonova Spectra automatically captures both 8-bin spectral and ultra-high definition spatial data simultaneously, without special setup or multiple protocols. The company said its system can harness up to 50 times more data than conventional CT through Nvidia computing technology.
Catherine Estrampes, president and CEO, U.S. and Canada at GE HealthCare, framed the clearance around rising pressure on clinicians: "As clinicians across the United States face rising volumes and increasing diagnostic complexity, technology must do more than capture images; it must simplify decision-making and strengthen performance across the enterprise. Photonova Spectra is designed to deliver rich clinical insights in every scan and help alleviate cognitive burden for care teams. With the U.S. 510(k) clearance, we are proud to now bring this innovation to U.S. healthcare systems and the patients they serve."
Jean-Luc Procaccini, president and CEO of Molecular Imaging and Computed Tomography at GE HealthCare, pointed to the depth of clinical collaboration behind the system. "Photonova Spectra reflects years of intentional design and close collaboration with clinicians, researchers and collaborators across the globe. From the earliest stages to today, we remain focused on building a system that addresses the practical realities of clinical practice while opening pathways for scientific advancement. The result is a photon-counting platform engineered for the needs of today's care teams, as well as the imaging challenges and research opportunities that will shape the future of CT."
For health systems already running GE equipment, the installation calculus is straightforward. The company indicates that Photonova Spectra is designed to be installed in existing CT environments with minimal modifications, specifically preserving the foundational mechanical, electrical, and workflow design used across the Revolution family of scanners. Workflow features include a one-scan approach that captures spectral and spatial data simultaneously, along with automated positioning and reconstruction tools, and the system is being evaluated at institutions including the University of Wisconsin-Madison and Stanford Medicine, where research is focused on image quality, workflow optimization, and potential clinical applications.
The FDA clearance quickly follows Photonova Spectra's debut at the Radiological Society of North America (RSNA) 2025 Annual Meeting, and the clearance is a result of the company's $5.1 billion innovation investment, leading to a wave of transformational products across the portfolio which combined are expected to drive 1-2% revenue growth.
Wall Street took notice of the timing. Citi Research analysts said the 510(k) clearance of Photonova followed years of anticipation, and the FDA nod positions GE HealthCare to show whether its claims about the advantages of its technology translate into market share. Citi Research analysts expect Photonova orders to ramp throughout 2026 and contribute to GE HealthCare sales next year, a forecast aligned with comments GE HealthCare CEO Peter Arduini made on an earnings call last month. Arduini said the order cycle is typically six to nine months, meaning that while some sales will be generated in 2026, 2027 will be the key year for new imaging products including Photonova.
The clearance also arrives alongside a broader acquisition push. Last week, GE HealthCare said it completed the acquisition of Intelerad, a medical imaging software provider, for a base price of $2.3 billion, signaling that the company is building out both the hardware and software sides of its imaging business simultaneously.
Know something we missed? Have a correction or additional information?
Submit a Tip

