Generate Biomedicines prices $400 million IPO to advance AI-designed asthma drug
Generate Biomedicines sold 25 million shares at $16 each to raise $400 million, funding late-stage trials of an AI‑engineered asthma antibody and sharpening debates over access and trial equity.

Generate Biomedicines priced its initial public offering at $16 a share, selling 25 million shares to raise roughly $400 million and listing on Nasdaq under the ticker GENB. The Somerville, Massachusetts company, founded by Flagship Pioneering in 2018, tapped Goldman Sachs and Morgan Stanley as joint bookrunners, with Piper Sandler, Guggenheim Securities and Cantor in the syndicate.
The offering will fund late-stage clinical work on the company’s lead program, GB-0895, an anti-TSLP antibody engineered with artificial intelligence to extend dosing intervals. Generate plans two Phase 3 trials in severe asthma that are expected to enroll about 1,600 patients combined, with full enrollment projected by the first half of 2028, and an ongoing Phase 1b study testing the antibody in chronic obstructive pulmonary disease. The company also intends to advance early cancer programs, including candidates labeled GB-4362 and GB-5267, into Phase 1 development while continuing platform work to generate additional programs.
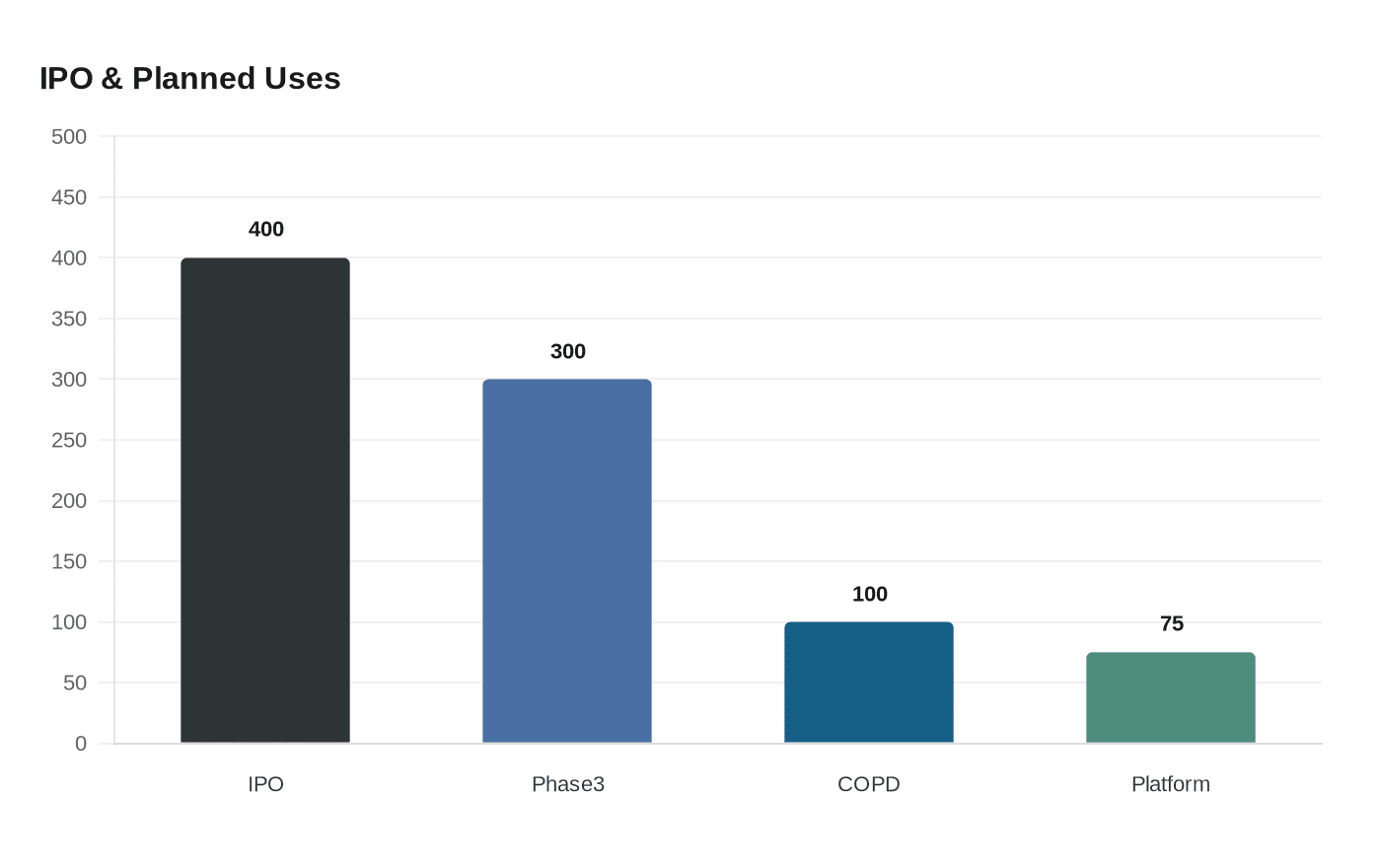
Generate is not yet profitable. Its prospectus shows roughly $31.9 million in revenue for the year ended Dec. 31, 2025, and a net loss of about $223 million. Company materials and analyst reporting set out an ambitious spending plan: about $300 million to complete the two Phase 3 asthma trials, an additional $100 million to finish COPD work and begin the next stages, and roughly $75 million for platform innovation and IND-enabling activities. Those estimates exceed the $400 million raised in this offering, underscoring that further capital will likely be required to execute the full development plan unless the underwriters exercise an overallotment option for up to 3.75 million additional shares.
For patients and public health systems, the IPO accelerates a transition from research to trials that could change management of severe asthma. An antibody engineered to last as long as six months between doses, if proven safe and effective, could reduce hospital visits and daily medication burdens for people with severe disease. At the same time, new biologics have historically entered the market with high price tags, raising immediate questions about affordability for patients on Medicaid and for communities where asthma prevalence and severity are disproportionately high.
Clinical trial design and enrollment will determine whether the benefits of an extended‑interval therapy reach the populations that need it most. Michael Rachlin, senior managing director at FTI Consulting, captured a broader market view: "We are likely in/entering a more discerning IPO marketplace where drug innovators with proven, later-stage assets/programs will have the necessary support to go public." That scrutiny extends to whether trial populations reflect racial, socioeconomic and geographic diversity and whether payers will secure pricing that preserves access.
Generate’s IPO is one of the largest recent biotech offerings, following several sizable new listings this year, and it highlights how capital markets continue to shape which biomedical innovations move fastest from platform to patient. As the company moves into registrational studies, policymakers and public health advocates will watch closely for signals about trial inclusivity, downstream pricing, and how publicly traded biotech financing affects equitable access to breakthrough therapies.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip

