Gleevec Approval in 2001 Launched Precision Medicine Era in Cancer Care
Gleevec turned a once-fatal leukemia into a treatable disease, and its anniversary exposes a harder truth: precision medicine changed survival before it fixed access.
Gleevec arrived as a proof of concept and a promise. When the Food and Drug Administration approved imatinib mesylate on May 10, 2001, it gave doctors a drug that targeted the Bcr-Abl tyrosine kinase created by the Philadelphia chromosome in chronic myeloid leukemia, and it gave patients something CML had rarely offered before: time.
The initial approval covered CML in blast crisis, accelerated phase, or chronic phase after failure of interferon-alpha therapy. The review took about two and a half months, a pace described at the time as one of the fastest drug approvals ever. In Washington, D.C., the announcement drew Health and Human Services Secretary Tommy Thompson and National Cancer Institute director Richard Klausner, underscoring how quickly the medicine moved from laboratory breakthrough to national health priority. Acting FDA Director Bernard Schwetz called Gleevec “one of the most effective drugs in years, perhaps ever” in cancer treatment.

That confidence was backed by early data that stunned oncologists. In Phase I testing, all 54 CML participants saw white blood cell counts return to normal within four weeks at doses of at least 300 mg daily. The response helped establish Gleevec, first developed as STI571, as one of the first widely recognized targeted cancer therapies and a landmark in the precision-medicine era that followed. Scientists including Brian J. Druker, Nicholas B. Lydon and Charles L. Sawyers helped drive the work that turned a molecular abnormality into a therapeutic target.
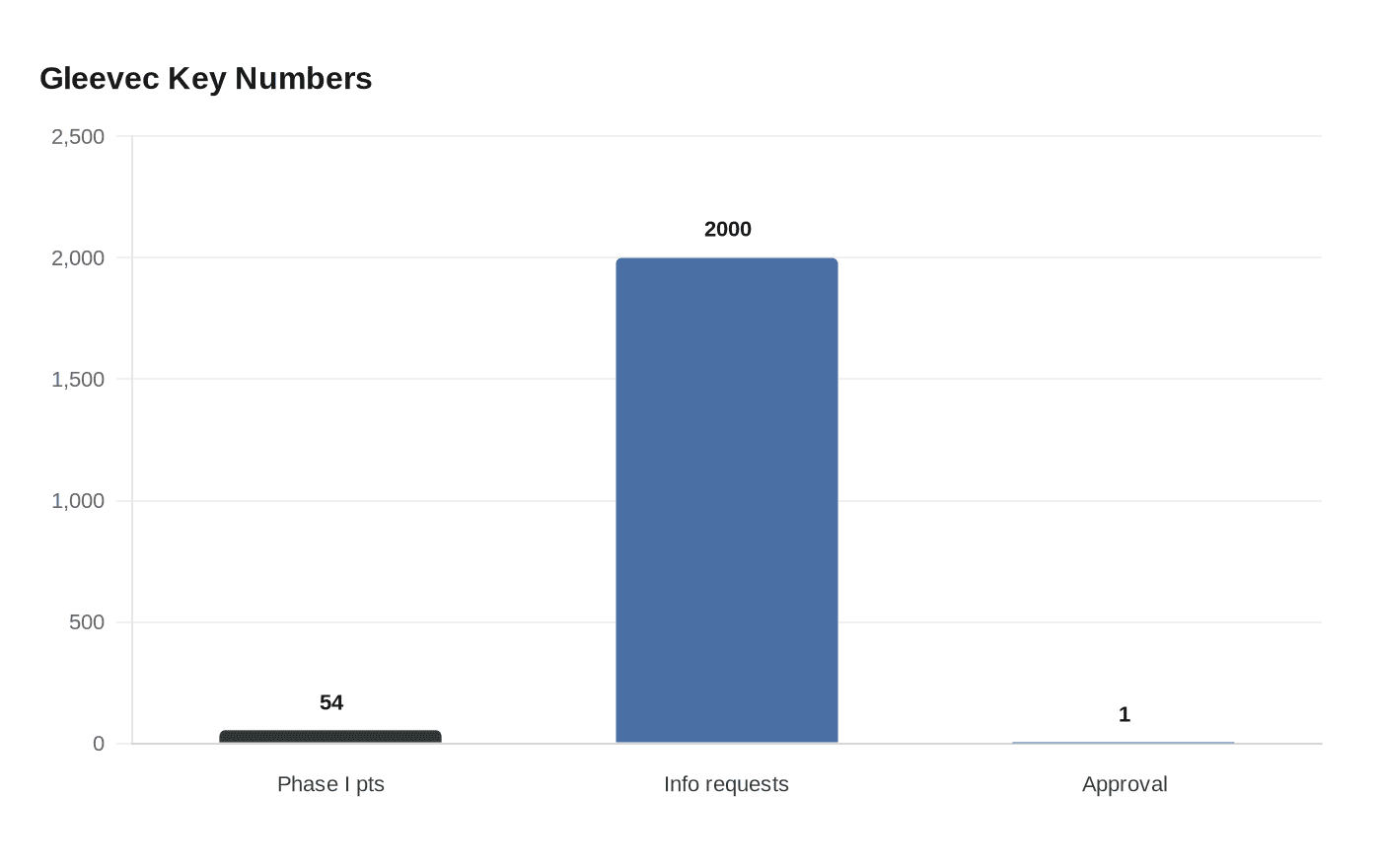
The public reaction showed how personal the advance felt. Novartis reportedly received more than 2,000 telephone requests for information within two days of the approval, a sign of the urgency surrounding a disease that had long carried grim expectations. National Cancer Institute material later described Gleevec as a landmark drug that helped launch targeted therapy and precision medicine, and National Institutes of Health material says newly diagnosed CML patients can now expect to live about 30 years after diagnosis, essentially a normal lifespan.
That survival gain is the triumph. The unresolved question is whether the system built around it can distribute the benefits evenly. Gleevec helped transform cancer care by showing that one mutation could be matched with one medicine, but it also helped define a new era in which scientific precision and financial strain advanced together. Twenty-five years later, the drug’s legacy is measured not only in lives extended, but in the continuing struggle to make such breakthroughs reachable for every patient who needs them.
Know something we missed? Have a correction or additional information?
Submit a Tip

