Harvard Scientists Found Nerve Overgrowth Blocks Scarless Skin Healing
A Harvard study published in Cell identified excessive nerve growth as a key barrier to scar-free wound healing, potentially reshaping regenerative medicine.

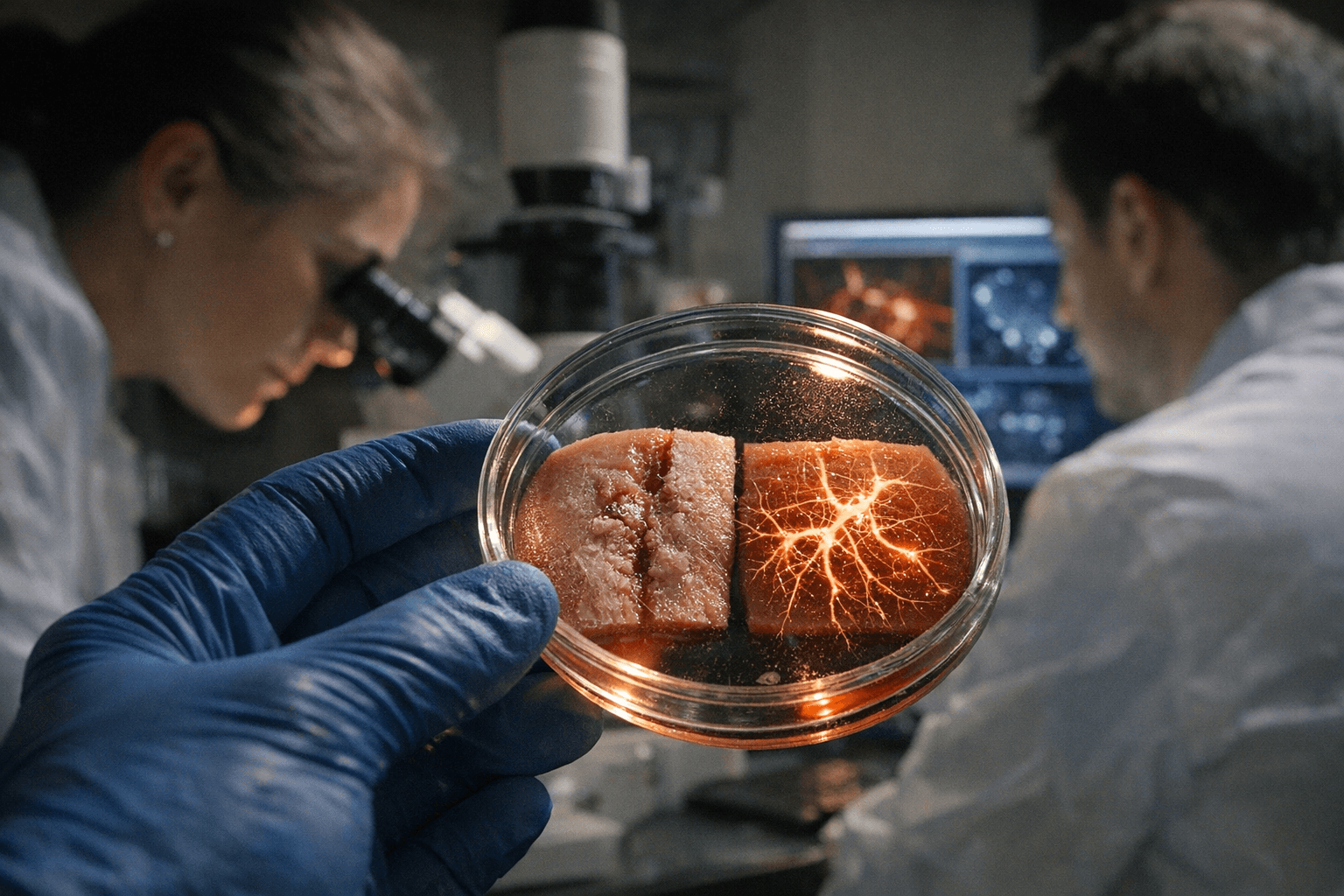
Professor Ya-Chieh Hsu's team at Harvard has pinpointed why human skin loses its ability to heal without scars after birth, identifying a process called hyperinnervation as the principal obstacle standing between current wound care and true organ-level regeneration.
The findings, published Thursday in the journal Cell, emerged from a systematic comparison of embryonic and postnatal wound healing. Embryonic skin, which heals without scarring, showed markedly different nerve growth patterns than skin healing after birth. In postnatal wounds, nerves proliferate excessively at the injury site, and that overgrowth, the study concluded, actively disrupts the regenerative process that would otherwise allow skin to restore itself completely, including hair follicles and other structural components lost in scarring.
The distinction matters enormously. Scar tissue is not simply healed skin. It lacks the complex architecture of normal tissue, including functional hair follicles, sweat glands, and the precise collagen organization that gives healthy skin its strength and flexibility. For the roughly 100 million patients in developed countries who acquire significant scars each year through surgery, injury, or burns, the difference between scarring and regeneration is a difference in long-term quality of life, sensation, and in severe cases, physical function.
Hyperinnervation had not previously been recognized as a central driver of this failure. The prevailing focus in regenerative medicine has centered on stem cell behavior, immune response, and growth factor signaling. Hsu's team reframed the question by examining the nervous system's role at the wound site, finding that nerve signals appear to push cells toward a repair pathway rather than a regenerative one.
The research carries particular weight for patients with burns covering large body surface areas, where the absence of regenerative healing creates permanent functional deficits and often requires repeated surgical interventions over years. It also has implications for conditions like diabetic foot ulcers, where impaired healing is already a significant driver of amputation.
From a health equity standpoint, access to advanced wound care is deeply uneven. Patients in lower-income communities and rural areas disproportionately face complications from poor wound healing, and any therapeutic advance that simplifies regenerative treatment could narrow that gap, though the path from laboratory finding to clinical application typically spans years of additional research and regulatory review.
The Cell paper positions nerve regulation as a new therapeutic target. If hyperinnervation can be selectively suppressed at wound sites without compromising the nervous system's essential role in healing, researchers may be able to recreate the conditions that allow embryonic skin to heal without a trace. That would represent a fundamental shift in how medicine approaches wounds, moving from managing damage to reversing it.
Hsu's lab has built its reputation around understanding skin stem cells and their microenvironments, and this finding extends that work into territory that could eventually reshape treatment protocols across surgery, burn care, and dermatology. The next challenge is translating a mechanistic discovery into a viable intervention, a process that will require testing whether modulating nerve growth in living patients produces the regenerative outcomes seen in the lab.
Know something we missed? Have a correction or additional information?
Submit a Tip

