HHS explored banning some antidepressants as Kennedy moves to curb SSRI use
Federal health officials weighed a ban on some antidepressants even as Robert F. Kennedy Jr. launched a push to cut SSRI use, putting millions of patients on edge.
Federal health officials explored whether they could ban some antidepressants in the SSRI class just as Robert F. Kennedy Jr. prepared a broader campaign to curb their use, a move that would have marked a sharp break in federal thinking about drugs taken by millions of Americans. The discussions centered on selective serotonin reuptake inhibitors, including sertraline, known as Zoloft, fluoxetine, known as Prozac, and escitalopram, known as Lexapro. HHS denied that any ban had been discussed, with spokesman Andrew Nixon calling the claim false.
Kennedy unveiled his MAHA Action Plan on May 4 at a summit focused on mental health and overmedicalization, saying the government would try to reduce psychiatric overprescribing rather than force patients off treatment. The plan called for education and outreach, research-to-practice efforts, billing-code guidance, and help for clinicians tapering patients off psychiatric drugs when clinically appropriate. The Centers for Medicare & Medicaid Services also issued guidance saying clinicians can be paid under Medicare for deprescribing-related care, while the Substance Abuse and Mental Health Services Administration was set to release a report on prescribing trends and a fact sheet for prescribers and patients later in May. Kennedy said, "If you are taking psychiatric medication, we are not telling you to stop."

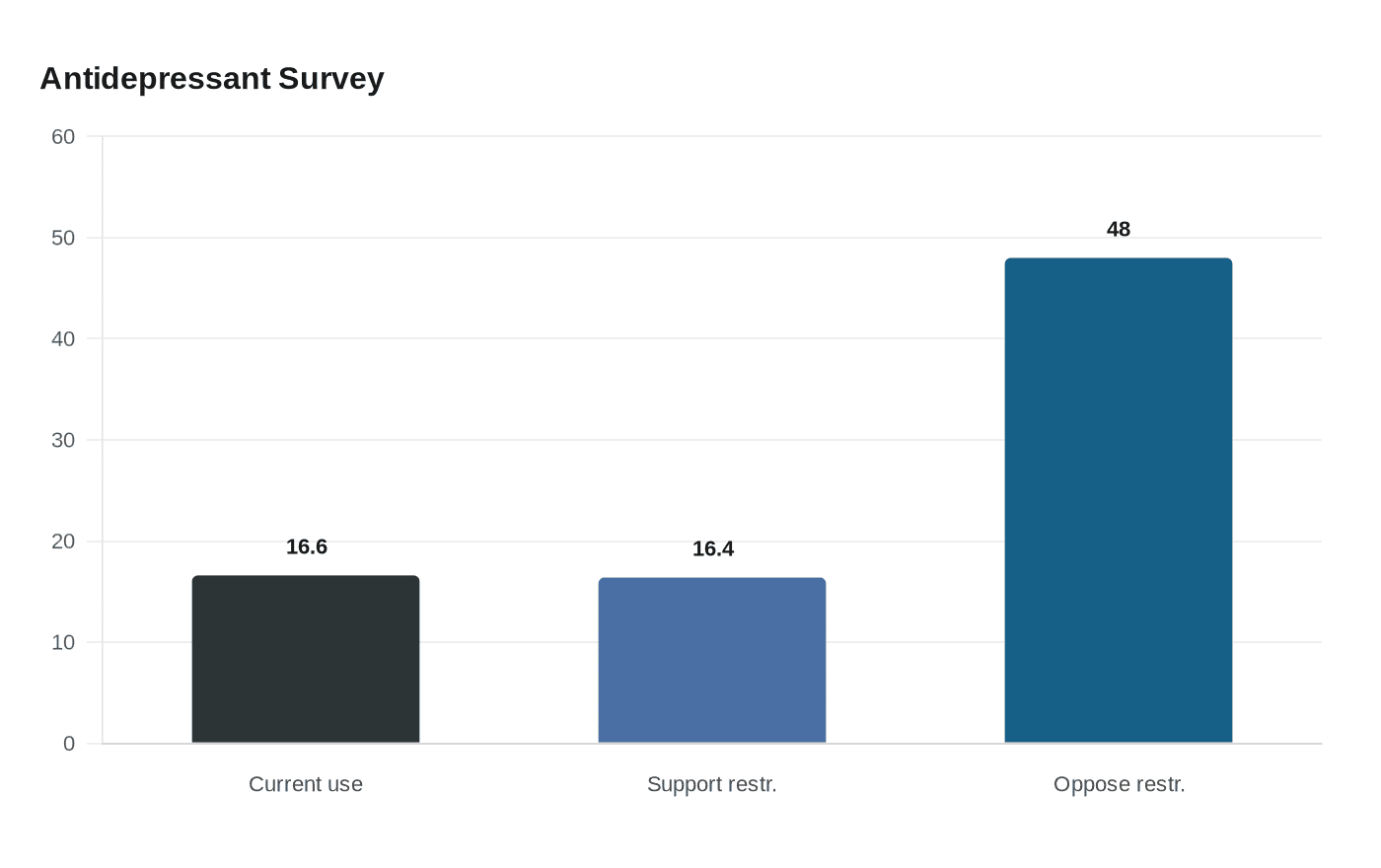
The policy stakes are high because antidepressants are widely used. A BMJ Mental Health survey of 30,810 adults found that 16.6% reported current antidepressant use, while 16.4% supported potential federal restrictions on prescribing and 48.0% opposed them. The study found that restrictive policies are unlikely to align with public sentiment and could worsen care inequities. The Food and Drug Administration says SSRIs are used to treat depression, anxiety and other mood disorders, and lists citalopram, escitalopram, fluoxetine, fluvoxamine, paroxetine, sertraline and vilazodone in the class.
Mental health specialists have warned that any federal campaign against SSRIs could confuse patients and deepen stigma around treatment. The American Psychological Association recommends psychotherapy and second-generation antidepressants, including SSRIs, for adult depression, while FDA labeling carries a boxed warning about increased suicidality risk in children, adolescents and young adults. American Psychiatric Association CEO Marketa Wills said she objected to a blanket overprescribing theory but wanted the association involved in any deprescribing guidelines. J. John Mann of the New York State Psychiatric Institute put the opposition more bluntly, saying, "Restricting use of these medications is not justifiable medically.
Know something we missed? Have a correction or additional information?
Submit a Tip

