IDEAYA, Servier cancer combo beats trial goal in rare melanoma
The combo cut progression risk in a rare eye cancer and raised fresh hopes for patients with few options. IDEAYA now faces the longer test of FDA review, pricing and access.

For patients with metastatic uveal melanoma, the result points to a potential new option in a disease where treatment choices remain thin and outcomes are often poor once the cancer reaches the liver. IDEAYA Biosciences and Servier said their darovasertib and crizotinib combination beat the main goal of a late-stage study, improving the time patients lived without their disease getting worse.
In the phase 2/3 OptimUM-02 trial, which enrolled 313 patients in the phase IIb/III portion, the combination produced a median progression-free survival of 6.9 months versus 3.1 months for investigator-chosen therapy. The hazard ratio was 0.42, with a 95% confidence interval of 0.30 to 0.59 and a p-value below 0.0001. The companies also reported an objective response rate of 37.1% versus 5.8%, including five complete responses in the darovasertib arm and none in the control arm.
IDEAYA said the study also showed an early trend toward improved overall survival, and that the combination was generally well tolerated with a manageable safety profile consistent with previously reported adverse events. The company plans to file a U.S. new drug application in the second half of 2026 and expects full OptimUM-02 data to be presented at a major medical conference next year. IDEAYA shares jumped sharply in premarket trading after the announcement, reflecting investor expectations that a positive readout in a rare cancer could eventually support approval and revenue.
The stakes are high because uveal melanoma is not ordinary skin melanoma. It starts in the uvea, the eye’s iris, ciliary body or choroid, and most cases begin in the choroid. The National Cancer Institute says intraocular melanoma is the most common eye cancer in adults, and metastatic disease is especially dangerous because it commonly spreads to the liver and carries a poor prognosis. Existing U.S. options are limited: the Food and Drug Administration approved tebentafusp-tebn in January 2022 for HLA-A*02:01-positive unresectable or metastatic uveal melanoma, and later cleared liver-directed melphalan in August 2023 for certain patients with unresectable hepatic metastases.
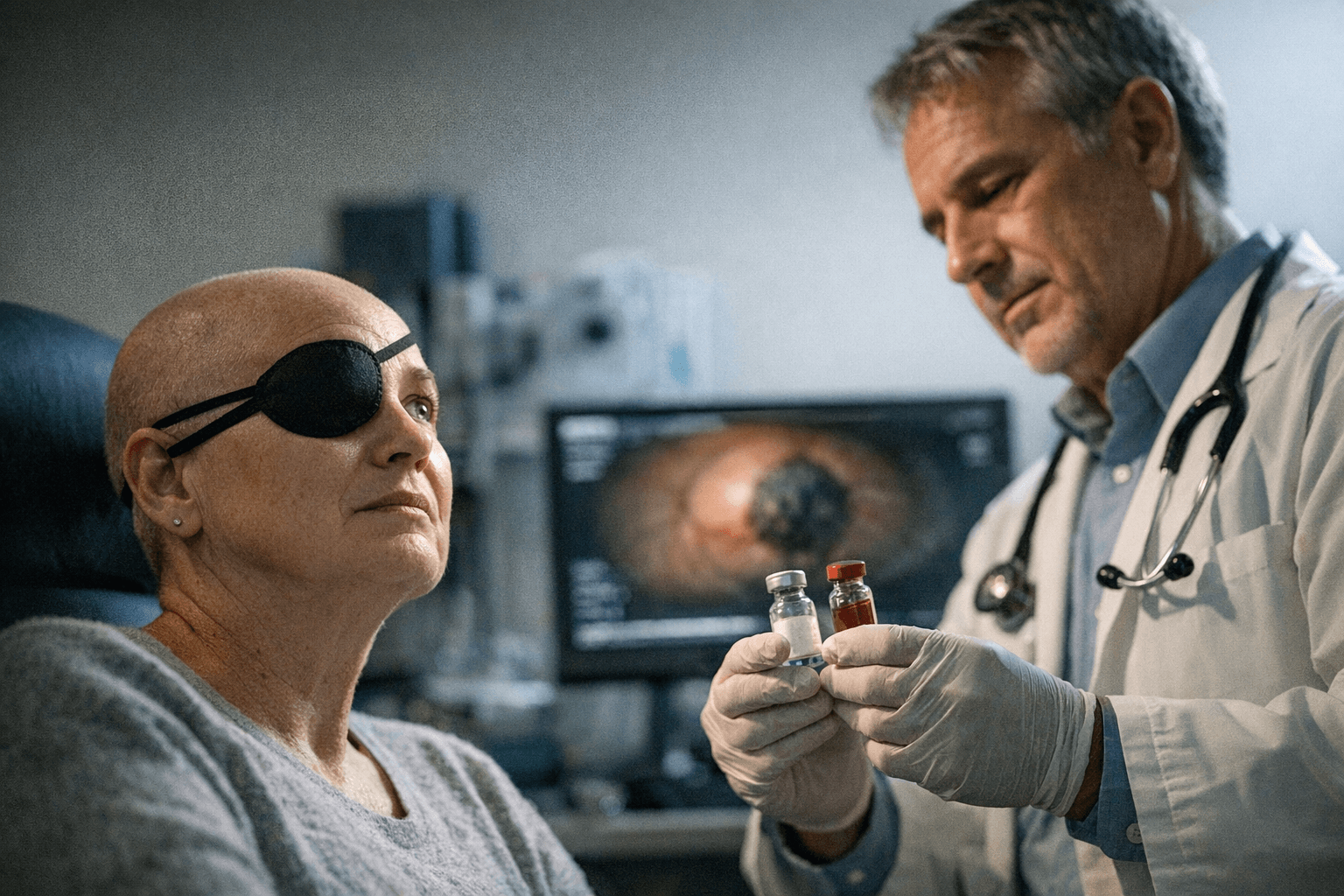
IDEAYA has also tried to build a broader franchise around the drug. On March 31, 2025, the FDA granted darovasertib Breakthrough Therapy designation in neoadjuvant primary uveal melanoma, after earlier data showed 82% ocular tumor shrinkage and 61% eye preservation. Chief medical officer Darrin Beaupre has said the company has been able to save eyes for patients who otherwise would need removal. Servier executive vice president of research and development Claude Bertrand called the result a very encouraging milestone, while IDEAYA chief executive Yujiro S. Hata said the data potentially offer a significant advance over currently available therapies.
Even with a strong trial, the path ahead is longer than a single headline. The company still needs to secure U.S. approval, then win pricing and coverage that can determine whether patients actually get the drug, especially in a rare cancer that is often treated at specialized centers.
Know something we missed? Have a correction or additional information?
Submit a Tip

