Medicare Semaglutide Price Agreements Could Expand Obesity Coverage While Cutting Costs
A JAMA study finds the $245/month BALANCE model price for semaglutide could offset coverage for up to 3.6 million more Medicare beneficiaries with obesity.
The arithmetic driving Washington's most expensive drug debate arrived in published form this week, and the numbers cut in two directions at once.
A research letter published April 2 in JAMA by Stacie B. Dusetzina, PhD, of Vanderbilt University, along with coauthors Grace Castle and Jihye Han, found that the $245-per-month semaglutide price negotiated under the Centers for Medicare and Medicaid Services BALANCE model could generate enough savings to offset the treatment costs of an additional 550,000 to 3.6 million Medicare beneficiaries eligible for expanded obesity-indication coverage. The wide range reflects how much the final arithmetic depends on assumptions about baseline use and whether further price reductions materialize beyond the model's initial terms.
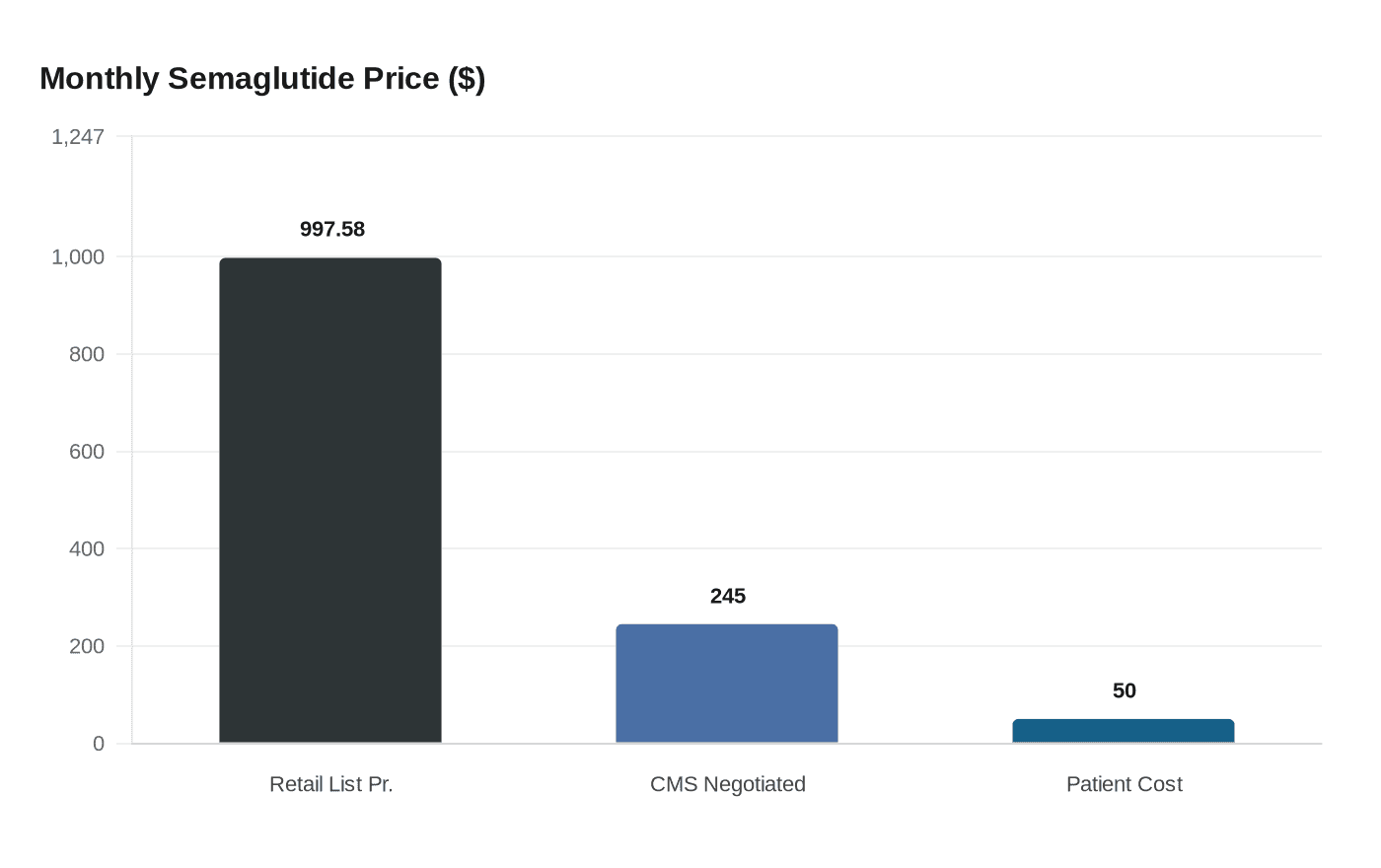
Under the BALANCE model, eligible Medicare beneficiaries would pay $50 per month for GLP-1 medications, against a retail list price for Ozempic, semaglutide's flagship brand, that sits at roughly $997.58 per monthly injection. That spread between list price and negotiated price is precisely where the savings headroom lives. If CMS captures most of it through the voluntary agreement mechanism, Dusetzina's team found the program could fund expanded access without requiring Congress to appropriate new dollars, at least within a modeled 2027 budget frame.
The stakes are measurable. Between 6 million and 14 million Medicare beneficiaries carry a diagnosis of obesity, a population currently blocked from coverage by a statutory prohibition on weight-loss drugs under Medicare Part D outside narrow cardiovascular and diabetes indications. The Congressional Budget Office estimated that covering anti-obesity medications for Medicare beneficiaries would cost roughly $35 billion over 10 years, with about $3 billion in health care savings from fewer hospitalizations and complications. The Dusetzina analysis layers a specific pricing lever onto that CBO frame, asking what happens to net spending if CMS locks in the BALANCE model price across semaglutide's entire Medicare utilization base before expanding the eligible population.
The BALANCE model, formally titled Better Approaches to Lifestyle and Nutrition for Comprehensive Health, will launch in Medicaid as early as May 2026 and in Medicare Part D in January 2027. Medicare Part D beneficiaries are expected to gain access to GLP-1s by July 2026 through a separate short-term demonstration serving as a bridge. That compressed timeline makes the modeling particularly consequential: the 2027 spending projection Dusetzina's team examined is not a distant hypothetical but a budget cycle already being priced by Medicare actuaries.
CMS negotiates standard key terms directly with each eligible manufacturer, and pricing agreements may include guaranteed rebates and manufacturer-provided access provisions. Novo Nordisk, which manufactures semaglutide, and Eli Lilly are the primary manufacturers whose participation shapes whether the model's $245 price holds or erodes at scale.
The JAMA letter arrived alongside a companion Viewpoint, "CMS BALANCE Model for Obesity: Implications for Patients and Clinicians," also published April 2, which examines how the model would reshape prescribing decisions and patient eligibility at the clinical level. Together, the two papers frame the same policy question from opposite ends of the transaction: what the program costs the federal government and what it means for the physician and patient trying to access it.
Whether voluntary price agreements can hold their savings projection as utilization scales remains the central uncertainty. The 550,000-to-3.6-million range in the JAMA letter is itself a signal that assumptions about adherence, discontinuation rates, and off-label demand carry as much weight as the negotiated price itself. A treatment population that churns quickly generates lower total drug spending but also fewer health outcomes, potentially undermining the downstream savings on hospitalizations and complications the CBO credited. The policy bet embedded in the BALANCE model is that a lower price, wider access, and sustained adherence can coexist, and that the net budget impact will remain manageable enough to survive the political scrutiny that $35 billion over a decade inevitably invites.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip