Moderna revenue triples on overseas COVID vaccine demand in first quarter
Moderna’s sales more than tripled to $389 million as overseas COVID orders dominated, but second-quarter guidance still pointed to a fragile recovery.

Overseas COVID vaccine orders gave Moderna a sharp first-quarter lift, but the company’s weaker second-quarter forecast showed how much of its business still depends on a pandemic franchise that is shrinking at home.
Moderna said revenue in the quarter reached $389 million, more than triple the year-earlier pace, with $78 million coming from the United States and $311 million from international markets. Roughly 80% of sales came from abroad, driven largely by deliveries under long-term strategic partnerships with government buyers in the United Kingdom, Canada and Australia. The company’s results underscore how far its vaccine business has shifted outside the U.S. as domestic demand remains clouded by changing federal policy.
The surge helped offset a $3.40 per-share GAAP loss, which was better than Wall Street had expected, but the quarter also included a $0.9 billion non-recurring litigation settlement charge. Investors still sent the stock down more than 2% in afternoon trading after Moderna said second-quarter revenue would fall to between $50 million and $100 million, well below the $130.6 million analyst estimate cited by the company. Moderna kept its outlook for as much as 10% revenue growth this year, but the guidance made clear that the near-term path remains uneven.
That uncertainty is especially sharp in the United States, where vaccine recommendations have become more politically and procedurally fraught under Health Secretary Robert F. Kennedy Jr. A federal court ruling in March froze work of the Advisory Committee on Immunization Practices, leaving the Centers for Disease Control and Prevention without a functioning advisory body to recommend new vaccines or expanded uses of existing ones. Without those recommendations, newly licensed vaccines may not be covered by insurers or included in federal programs, limiting the domestic market Moderna needs to stabilize.
Moderna is trying to prove that its mRNA platform can outgrow COVID. The company is investing in respiratory vaccines, including a combination COVID-flu shot, while also pushing into oncology and rare diseases. Stéphane Bancel said the company expects several additional approvals around the world this year, including for its seasonal flu vaccine, which would become Moderna’s fifth approved product. In Europe, the company won a key endorsement when the European Medicines Agency recommended its mCombriax combination shot for adults 50 and older on February 27, based on a study of 8,000 people; the European Commission approved it on April 21. Moderna says mCombriax is the world’s first flu-plus-COVID combination vaccine to receive marketing authorization, and its mRNA-1083 filings remain under review in Canada and Australia.
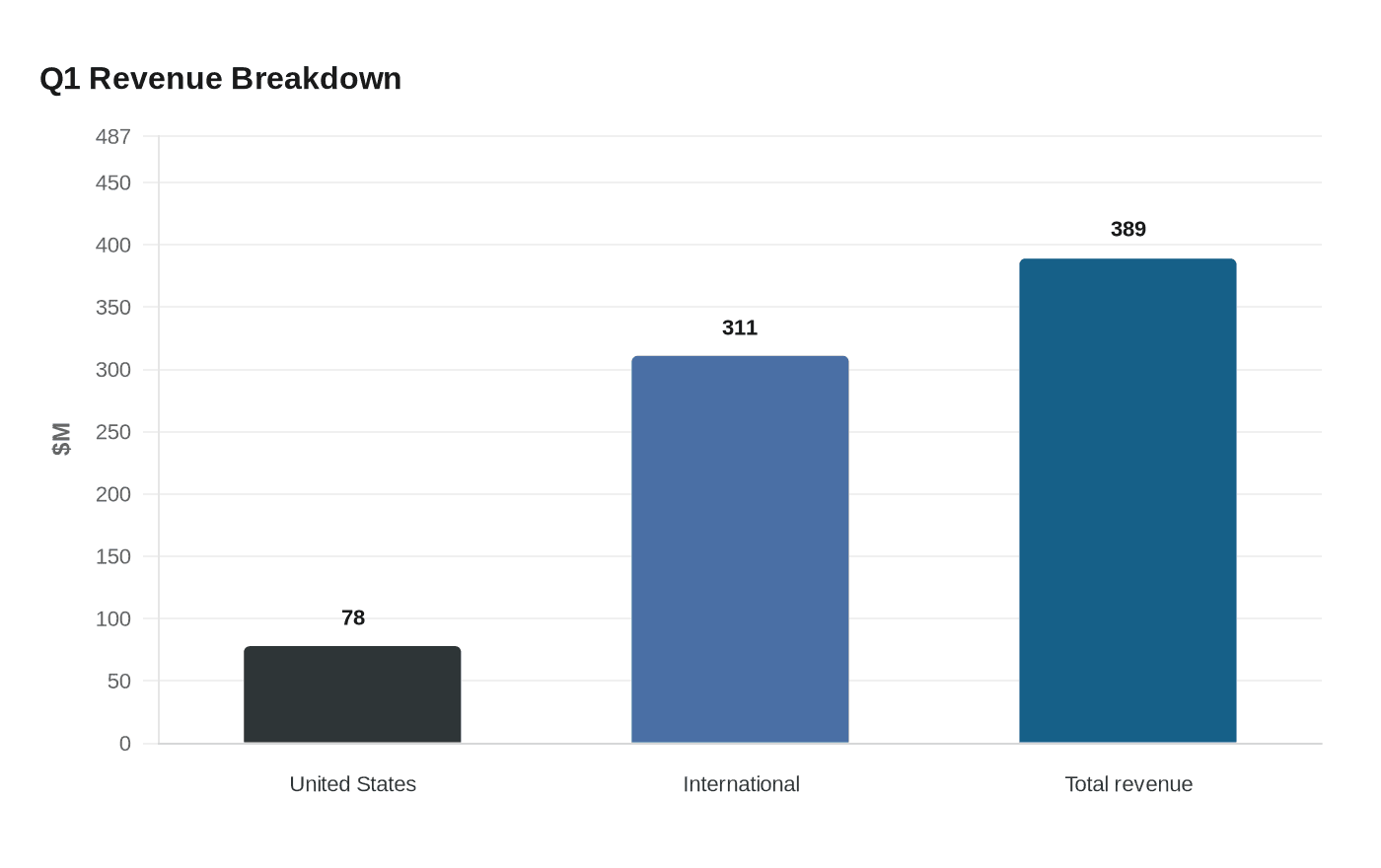
The broader test for Moderna is not whether overseas COVID demand can produce another quarterly pop. It is whether that demand can finance a diversified portfolio before the pandemic revenue that built the company fades further.
Know something we missed? Have a correction or additional information?
Submit a Tip

