Modular Medical Wins FDA Clearance for Pivot Tubeless Insulin Patch Pump
Modular Medical's Pivot patch pump won FDA clearance, targeting the 70% of insulin-dependent adults who avoid pumps due to cost and complexity.
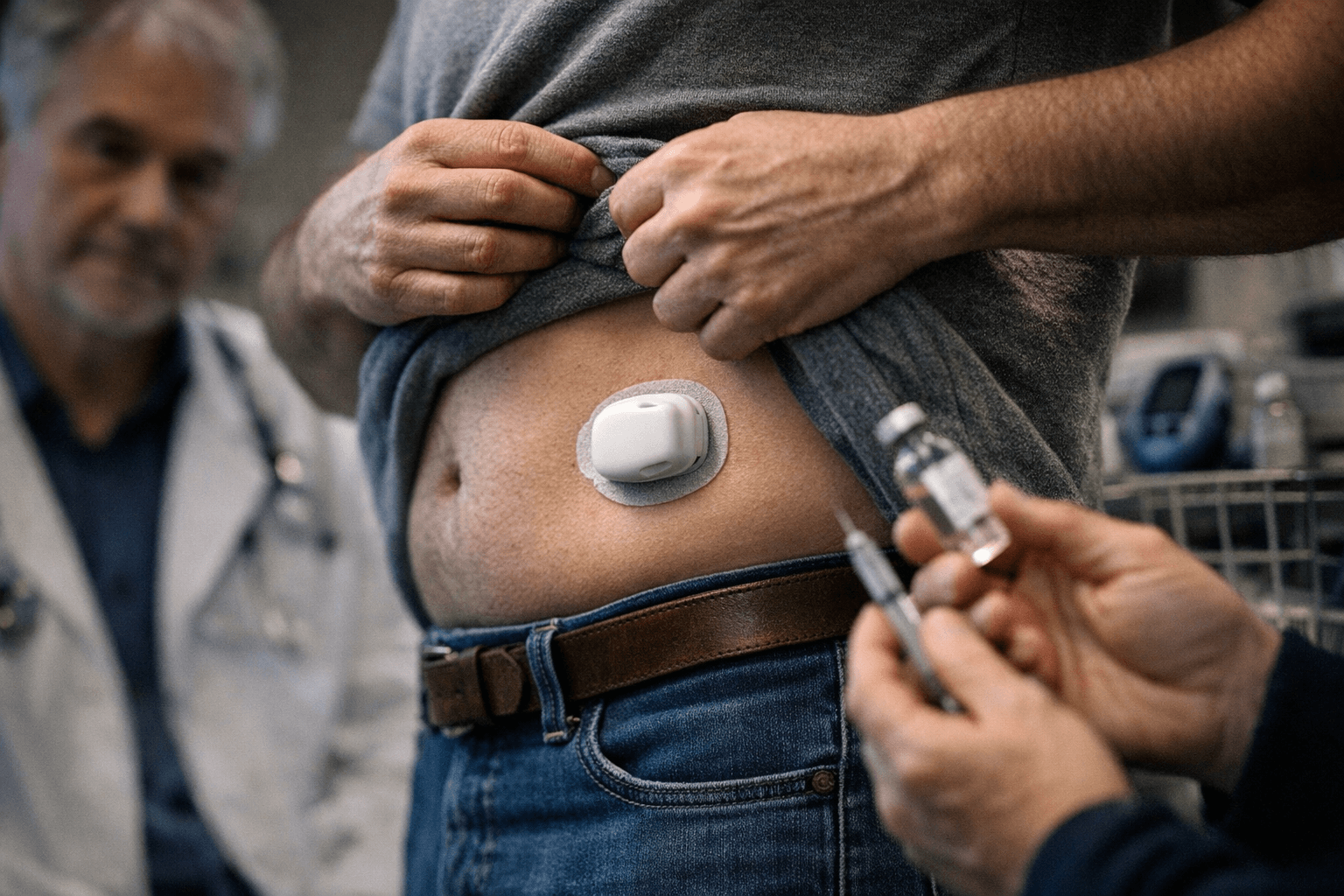
Modular Medical Inc. secured 510(k) clearance from the U.S. Food and Drug Administration for its Pivot tubeless insulin patch pump, removing the primary regulatory hurdle to bringing a device designed for the millions of diabetes patients who have resisted conventional pump therapy to U.S. commercial markets.
The San Diego company announced the clearance Wednesday, with CEO Jeb Besser describing it as a "major de-risking event" and framing Pivot as a direct answer to why roughly 70% of insulin-dependent adults remain on multiple daily injections rather than switching to pump therapy. "FDA clearance for Pivot represents years of focused innovation to deliver a truly differentiated tubeless patch pump," Besser said. "By addressing the key barriers, complexity, bulkiness, and high cost, we believe we are positioned to convert millions still on injections to pump therapy."
The device takes a two-part form factor: a reusable electronic module paired with a disposable component housing a 3 mL reservoir and a replaceable battery. Unlike conventional patch pumps that require constant wear, Pivot can be detached for activities such as showering or sports, which Modular Medical argues removes one of the most cited friction points among the population it calls "almost-pumpers." Smartphone connectivity supports bolus delivery and remote monitoring.
Manufacturing validation lots are already underway, and the company set initial production capacity to support approximately 6,000 users. First customer shipments are targeted by the end of the second quarter of 2026. Modular also outlined a software development roadmap that includes variable bolus functionality, enhanced alarm systems, and eventual compatibility with automated insulin delivery platforms, the closed-loop technology increasingly considered the standard of care for type 1 diabetes management.
International expansion is already in planning. The company said it is pursuing CE marking for European markets, with that approval process expected to conclude in late 2026 or early 2027.
Besser characterized the addressable market in the billions of dollars, pointing to the large share of the insulin-dependent population that has never adopted pump therapy. The reusable-plus-disposable architecture is central to Modular's cost argument: by separating the expensive electronics from the consumable components, the company believes it can price Pivot below legacy pump systems and reach patients without robust insurance coverage.
Still, FDA clearance marks a threshold rather than a finish line. Reimbursement coverage from major payers, uptake among endocrinologists and certified diabetes educators, and real-world clinical data on outcomes including HbA1c improvement and hypoglycemia reduction will determine whether Pivot converts its regulatory win into commercial traction. The insulin pump space is populated by entrenched competitors, and payer decisions will hinge on demonstrated efficacy and cost-effectiveness relative to existing options. Investors and health systems will be watching early post-market performance and any pricing and coverage announcements closely as shipments begin this summer.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip

