mRNA vaccines show new promise against deadly pancreatic cancer
A pancreatic cancer patient’s experimental mRNA shot helped revive a platform once battered by vaccine backlash, as federal funding and early trial data drew new attention.

Vita Sara Blechner’s diagnosis in 2020 showed how little margin pancreatic cancer still allows. The middle school librarian was rushed through emergency-room scans before learning she had one of the deadliest cancers, a disease that leaves only about one in four patients alive a year after diagnosis and roughly one in ten alive two years later. That grim math has made even small gains in mRNA research matter, because the platform that powered Covid-19 vaccines is now being tested as a way to teach the immune system to recognize tumors.
The comeback case for mRNA is being built on more than hope. In March 2026, the National Cancer Institute and the Foundation for the National Institutes of Health launched a partnership aimed at raising nearly $200 million for cancer vaccine clinical trials, including mRNA-based approaches for patients at high risk of tumor recurrence. The funding push underscored a broader shift in the field after a difficult stretch marked by political backlash against pandemic-era vaccines, uncertainty over support, and skepticism that mRNA could do much beyond fighting viruses. Researchers have argued for years that the platform is adaptable, and companies such as BioNTech were already pursuing cancer programs before Covid vaccines made the technology familiar worldwide.
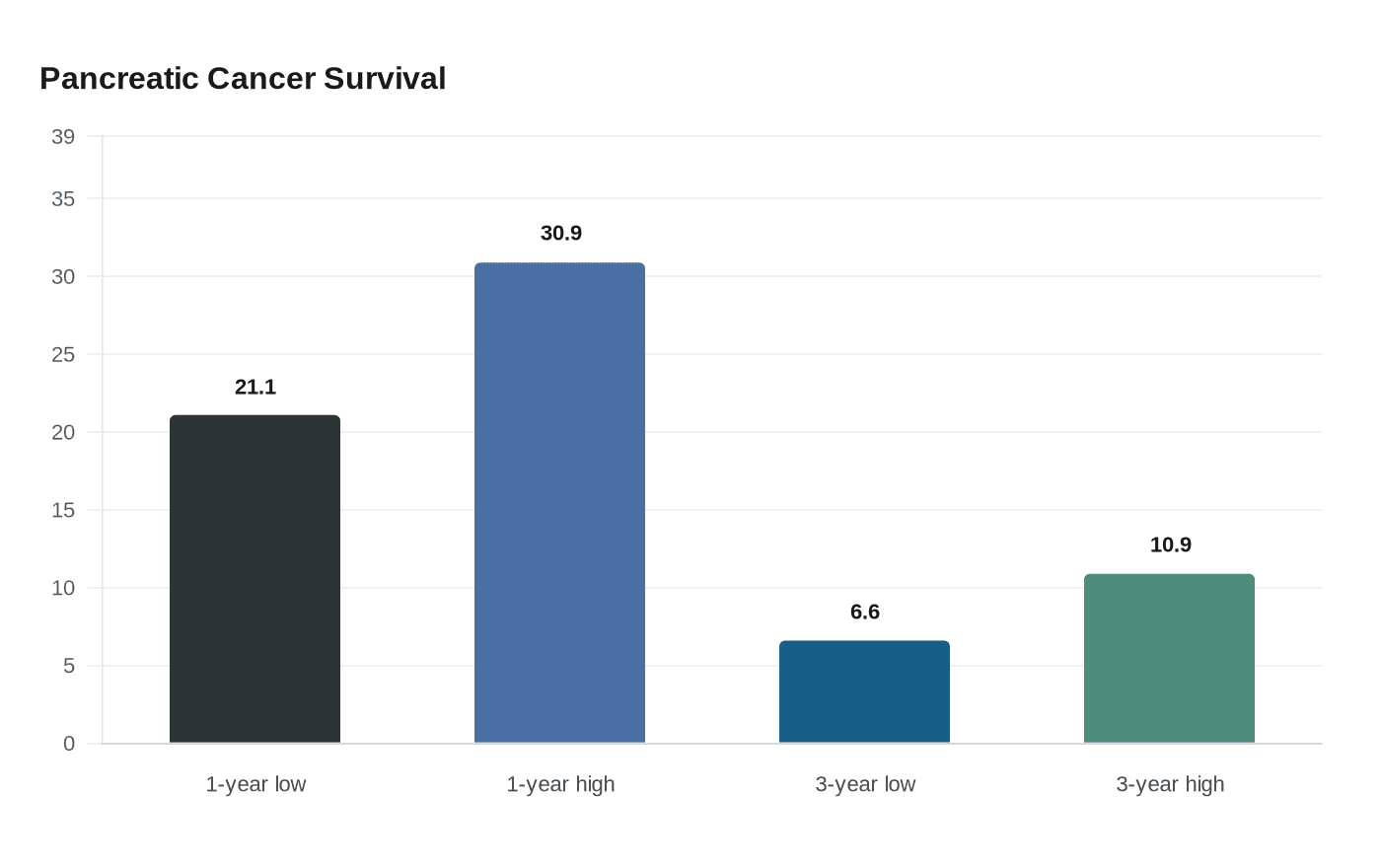
The strongest early signal has come from Memorial Sloan Kettering Cancer Center’s phase 1 pancreatic cancer study of autogene cevumeran, an individualized neoantigen vaccine built on uridine mRNA-lipoplex nanoparticles. Sixteen patients received the vaccine after surgery, alongside chemotherapy and a checkpoint inhibitor. Eight of the 16 showed vaccine-induced tumor-specific immune responses. Extended follow-up published in Nature found that, at a median 3.2 years, responders had prolonged recurrence-free survival, with the median not reached in responders versus 13.4 months in non-responders. Memorial Sloan Kettering later said the trial still showed evidence of a robust vaccine-activated T-cell response at three years, and in a 2025 update the institute said some responders were still alive years later.
Pancreatic cancer’s toll helps explain why these findings are drawing so much attention. SEER estimated that 107,988 people in the United States were living with the disease in 2022, while NIH says it accounts for about 3% of new cancer diagnoses but 8% of cancer deaths. A multi-country study found one-year survival ranging from 21.1% to 30.9% and three-year survival from 6.6% to 10.9%. For mRNA to move from promise to proof, researchers will need larger trials, durable survival gains, and results that hold up across centers and tumor types. Anything less would be another burst of hope, not a new standard of care.
Know something we missed? Have a correction or additional information?
Submit a Tip

