Novo Nordisk’s CagriSema misses non-inferiority; shares plunge 16%
Novo Nordisk’s REDEFINE-4 topline showed CagriSema fell short versus tirzepatide, triggering a 12-16% stock slide and a surge in trading volume.

Novo Nordisk said topline results from the Phase 3 REDEFINE-4 head-to-head trial showed CagriSema produced substantial weight loss but “did not achieve its primary endpoint of demonstrating non‑inferiority on weight loss for CagriSema compared to tirzepatide after 84 weeks,” a company statement reported by multiple outlets. The announcement sparked an immediate market reaction, with shares tumbling in Copenhagen and New York and trading volume spiking.
Clinical accounts of the trial’s efficacy differ across published reports. Bloomberg and STAT cited Novo Nordisk figures showing patients treated with a standard dose of CagriSema lost 20.2% of body weight over 84 weeks, versus 23.6% for Eli Lilly’s tirzepatide. By contrast, Motley Fool and The Guardian reported higher numbers for an adherent population: 23% average weight loss on CagriSema against 25.5% for tirzepatide, with The Guardian noting the study involved 809 people and that the 23% figure applied “if all people adhered to treatment.” STAT described REDEFINE-4 as open-label. None of the reports explained the differing analytic populations or dosing labels in detail.
Investors seized on the headline result. Motley Fool reported Novo Nordisk closed at $39.63, down 16.43% on Feb. 23, with trading volume reaching 94.9 million shares, about 343% above its three-month average. CNBC and Bloomberg cited intraday declines of roughly 12% to 15% in Copenhagen, with a reported low of 256 Danish kroner. Eli Lilly shares rose in response, gaining between about 3.5% and 4.9% across outlets in premarket and intraday trading. Deutsche Bank downgraded Novo from Buy to Hold, according to Motley Fool.
Novo’s chief scientific officer defended the program’s rationale and future plans. Martin Holst Lange said CagriSema “has the potential to be the first GLP‑1/amylin‑combination product to reach the market for people living with obesity, documenting that cagrilintide adds to the existing benefits of semaglutide and offers clinically meaningful additive weight loss effects superior to what has been observed with GLP‑1 biology alone,” and that further trials would “assess the full weight‑loss potential of CagriSema,” a CNBC report quoted.
Analysts framed the result as a strategic setback for Novo Nordisk as competition intensifies. Søren Løntoft Hansen, senior analyst at AL Sydbank, said: “This is something of a swing and a miss.” He added: “It is difficult to assess whether this data will influence Novo Nordisk’s decision to launch Cagrisema on the market.” Bloomberg and other outlets noted the setback comes as Novo grapples with rising competition, pressure on U.S. pricing, and looming loss of exclusivity for Wegovy and Ozempic.
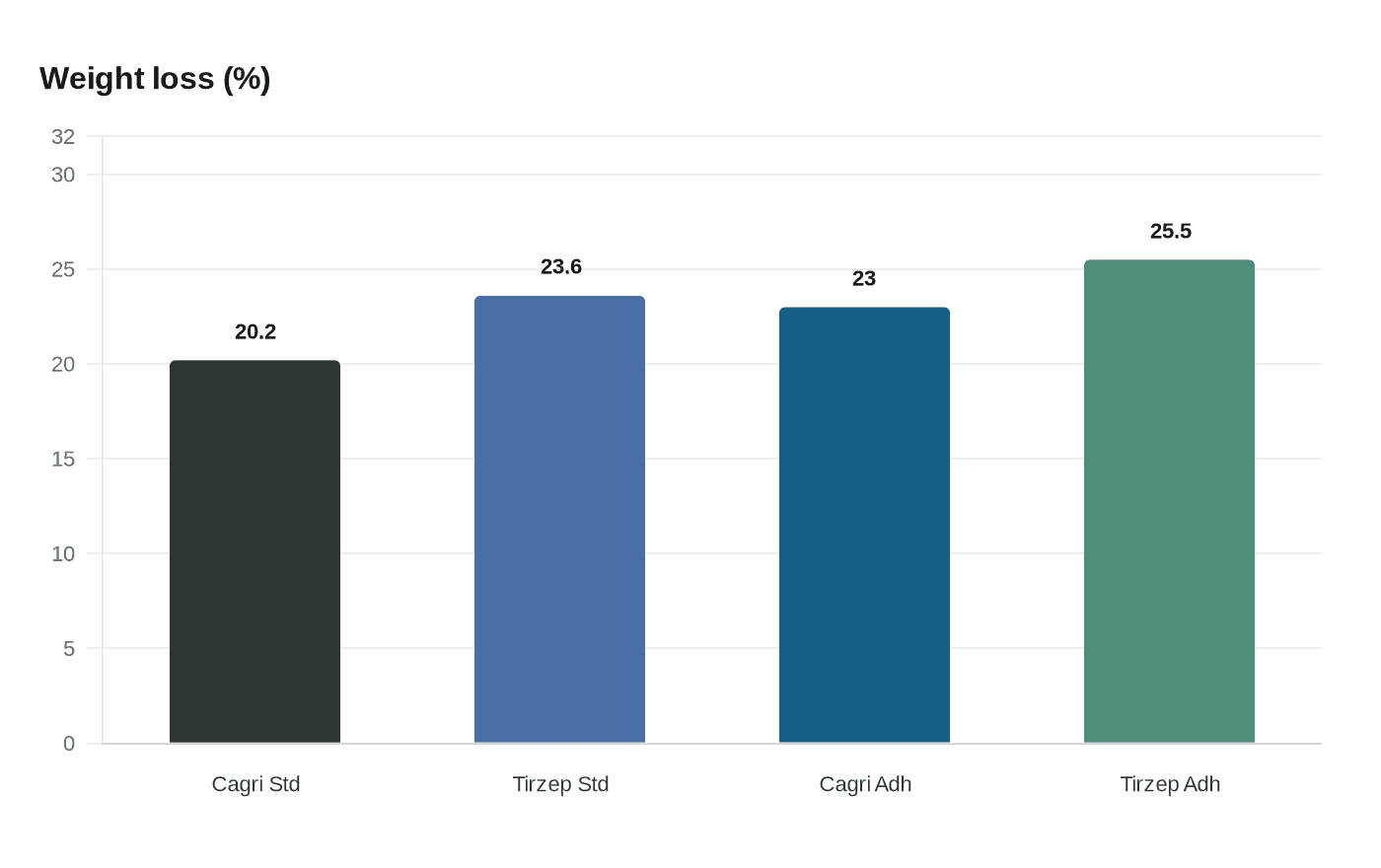
Market implications go beyond headline share moves. A failed non-inferiority result complicates Novo’s plans to extend its dominant position in obesity treatments and could preserve pricing and market share advantages for Lilly’s tirzepatide franchise, including Zepbound. The discrepancy in reported weight-loss figures also raises an immediate need for the company to release detailed topline documents clarifying analytic populations, dose cohorts, safety and adherence data, and any planned changes to development or commercialization strategy.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip

