Rare ALS drug shows breathing, strength gains in some patients
In some ALS patients, tofersen not only slowed decline but restored grip strength and breathing, a rare gain in a disease that usually worsens relentlessly.

For a small slice of ALS patients, the signal was startling: breathing and strength did not just hold steady with tofersen, they improved. In long-term follow-up, some people with SOD1-linked ALS regained functions they had already lost, a rare result in a disease that usually moves in one direction.
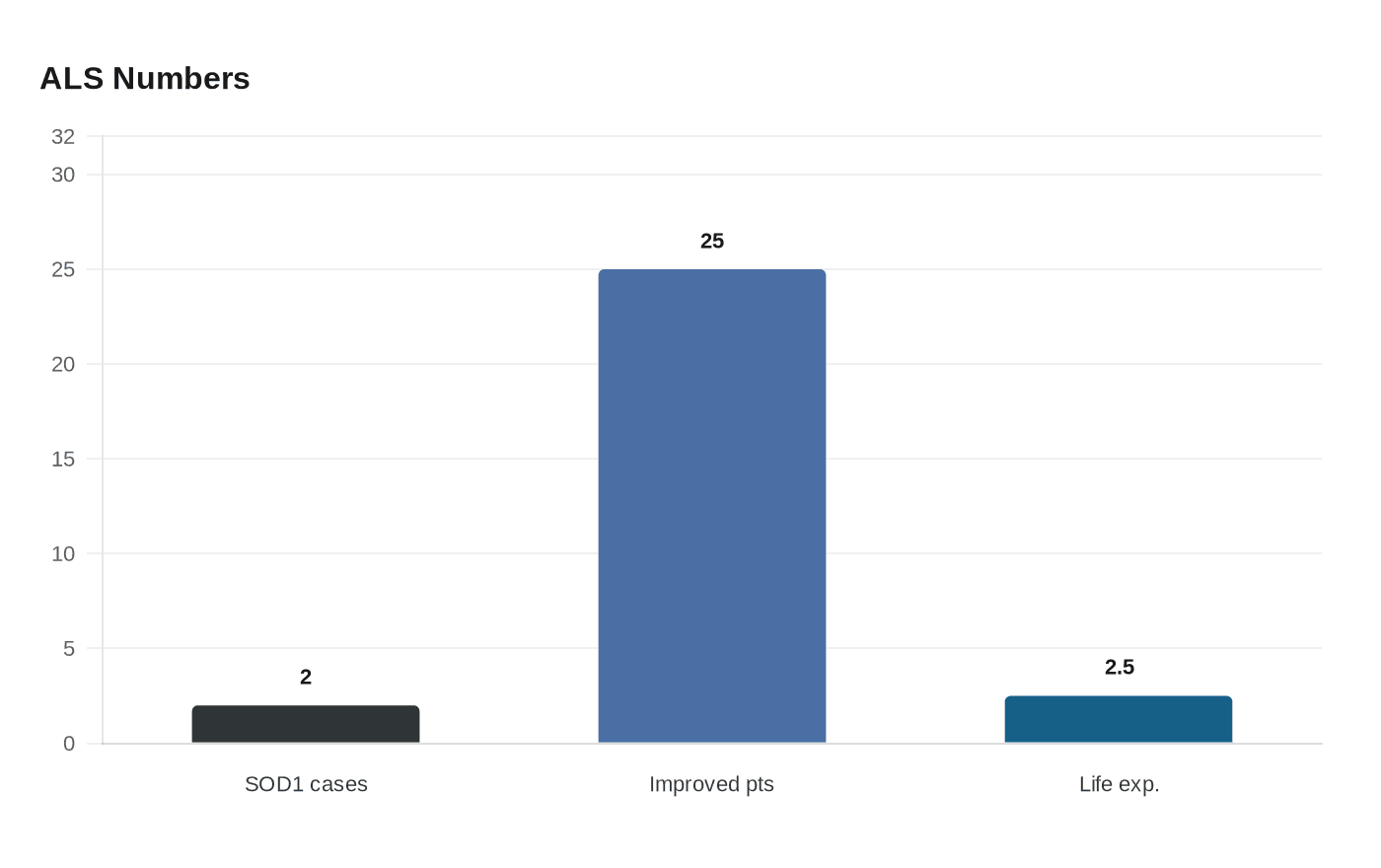
That matters well beyond this drug. Tofersen, sold as Qalsody, is the first FDA-approved treatment to target a genetic cause of ALS, and its latest data offer unusually concrete evidence that a fatal neurodegenerative disease may be not only slowed but partly reversed in some patients. Researchers at Washington University in St. Louis said roughly one-quarter of participants in one study group experienced stabilization and even functional improvement in grip strength and respiratory function over more than three years of follow-up.
The treatment is aimed at ALS caused by mutations in the SOD1 gene, a tiny subgroup that accounts for about 2% of ALS cases. Biogen said about 330 people in the United States had this ultra-rare form of the disease when the drug won accelerated approval from the U.S. Food and Drug Administration on April 25, 2023. The approval rested on lowering neurofilament light chain, a blood marker of nerve-cell damage, rather than on definitive proof at the time that patients lived longer or functioned better.
The new long-term VALOR and open-label extension data pointed in the direction regulators, doctors and patients had hoped for. Earlier treatment was associated with less decline in function, strength, respiratory capacity and quality of life, along with fewer death-equivalent events. That is especially notable in SOD1-ALS, where average life expectancy is about two to three years from symptom onset and progression is typically relentless.
The findings do not erase the limits. Tofersen applies only to a very small genetic subgroup, and the durability of the benefit still needs to be proven over longer periods. Continued approval depends on confirmatory evidence, and the ongoing ATLAS study is testing tofersen in presymptomatic SOD1 mutation carriers as part of that requirement.
Still, the broader significance is hard to miss. The ALS Association called the 2023 approval the first ALS therapy cleared under the FDA’s accelerated approval pathway and an important endorsement of antisense technology. For Cambridge, Massachusetts-based Biogen and Ionis Pharmaceuticals, the company behind the antisense approach, the drug has become a test case for whether genetic targeting can change the course of diseases once thought to be uniformly unstoppable.
Know something we missed? Have a correction or additional information?
Submit a Tip

