Sanofi beats profit and revenue forecasts on strong Dupixent demand
Dupixent again powered Sanofi past forecasts, but the bigger test is whether Belén Garijo can broaden growth before concentration risk deepens.

Dupixent again did the heavy lifting for Sanofi, lifting first-quarter sales above forecasts and giving incoming chief executive Belén Garijo a stronger hand as she prepares to take over at the end of April. The French drugmaker said sales reached 10.509 billion euros in the three months to March 31, up 13.6% at constant exchange rates, while business operating income rose to 2.967 billion euros.
The standout was Dupixent, Sanofi’s asthma and eczema treatment, which climbed 30.8% to about 4.2 billion euros and moved back above the 4 billion-euro quarterly mark. That kind of growth keeps the company’s earnings engine running, but it also sharpens the concentration risk that has followed Sanofi into the leadership transition. One drug is still doing a disproportionate amount of the work for a company that needs to prove its next phase will not depend on a single franchise forever.
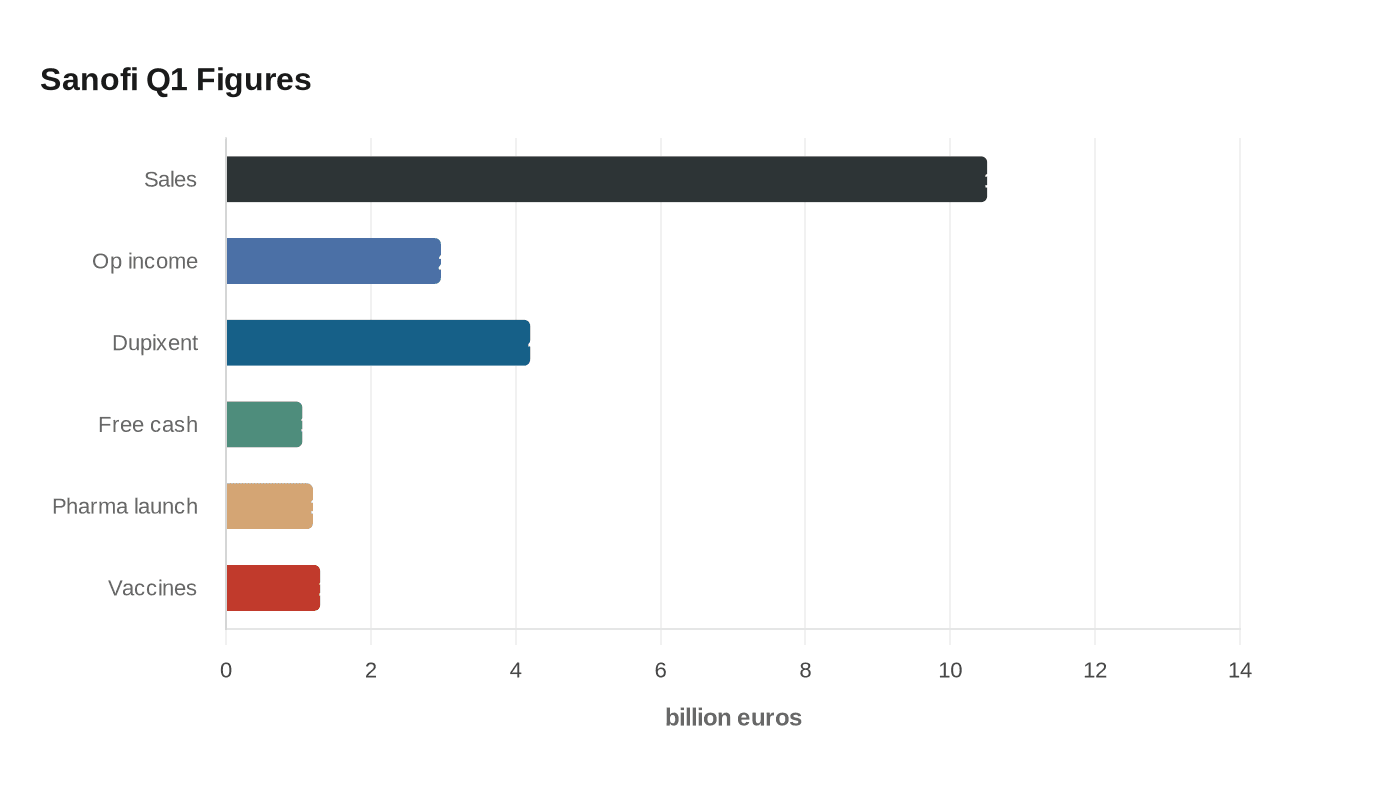
Sanofi said business earnings per share came in at 1.88 euros, while IFRS earnings per share were 1.34 euros. Free cash flow totaled 1.054 billion euros. The company reaffirmed its 2026 guidance for high single-digit sales growth at constant exchange rates and said earnings per share should grow slightly faster than sales, a signal that management believes the current operating momentum can continue.
The quarter also showed signs that Sanofi is trying to widen its base beyond Dupixent. Pharma launches sales increased 49.6% to 1.2 billion euros, led by Ayvakit, ALTUVIIIO and Sarclisa. Vaccines sales rose 2.1% to 1.3 billion euros, helped by Heplisav-B. Sanofi also completed the Dynavax acquisition and finished 921 million euros of its 1 billion-euro share buyback program, a reminder that capital allocation remains part of the story even as the pipeline is still being rebuilt.
That pipeline matters because Sanofi enters the Garijo era under pressure to show it can create the next growth engine. The company reported five regulatory approvals, all in immunology, along with positive phase 3 data for venglustat in GD3 and phase 2 data for lunsekimig in respiratory disease. Those are useful steps, but they do not yet replace the scale or certainty that Dupixent provides.
Sanofi shook up its leadership in February when it removed Paul Hudson and named Garijo to succeed him after the April 29 annual general meeting, with Olivier Charmeil serving as interim chief executive. Shares rose nearly 3% in morning trading after the results, suggesting investors welcomed the beat and the guidance, but the next phase will be judged on a harder question: whether Sanofi can keep growing if Dupixent eventually stops carrying so much of the load.
Know something we missed? Have a correction or additional information?
Submit a Tip

