Small Swedish medtech SciBase wins FDA label expansion for Nevisense
SciBase says the FDA approved a supplemental labeling change for Nevisense on March 2, 2026, expanding its use in the United States.

SciBase Holding AB announced on March 2, 2026 that the U.S. Food and Drug Administration approved a supplemental labeling change expanding the use of Nevisense in the United States. The company, headquartered in Sundbyberg near Stockholm and listed on Nasdaq First North since June 2, 2015, framed the action as a step toward broader point-of-care deployment for its electrical impedance spectroscopy device that integrates an AI classifier.
Nevisense received Pre-market Approval in June 2017 and is classified by the FDA as a Class III device, a designation the company has described as reflecting the diagnostic risk profile for melanoma and a lack of predicate devices. SciBase investor materials state that “Nevisense method and its AI classifier are scientifically proven through a prospective (forward looking) clinical study with almost 2,000 patients and 2,400 lesions, which is the world’s largest clinical study of its kind.” The company also says, “At present, SciBase is the only Company with approved products that uses AI and EIS for the detection of skin cancer. Nevisense is the only point-of-care product with FDA approval for melanoma detection available in the United States.”
The supplement is a regulatory milestone for a small medtech competing in a field dominated by larger device makers and imaging platforms. SciBase has previously emphasized the demands of the PMA process, calling it “the rigorous Pre-market approval (PMA) process that aims to ensure the highest-quality of clinical results and product.” The company reports partnerships and recognition that could help scale adoption, including a collaboration with Mayo Clinic on pigmented lesion digital workflows and inclusion of Nevisense in updated German S1 imaging guidelines.
Clinically, broader label permissions for an AI-driven, point-of-care melanoma detection tool could reshape workflows in primary care and dermatology clinics. Proponents argue that widening access to diagnostic tools at the point of care may improve early detection of melanoma, particularly in communities with limited specialist access, and reduce unnecessary referrals and biopsies. The device’s prospective study sample of nearly 2,000 patients and 2,400 lesions is a notable data point for policymakers weighing adoption and reimbursement.
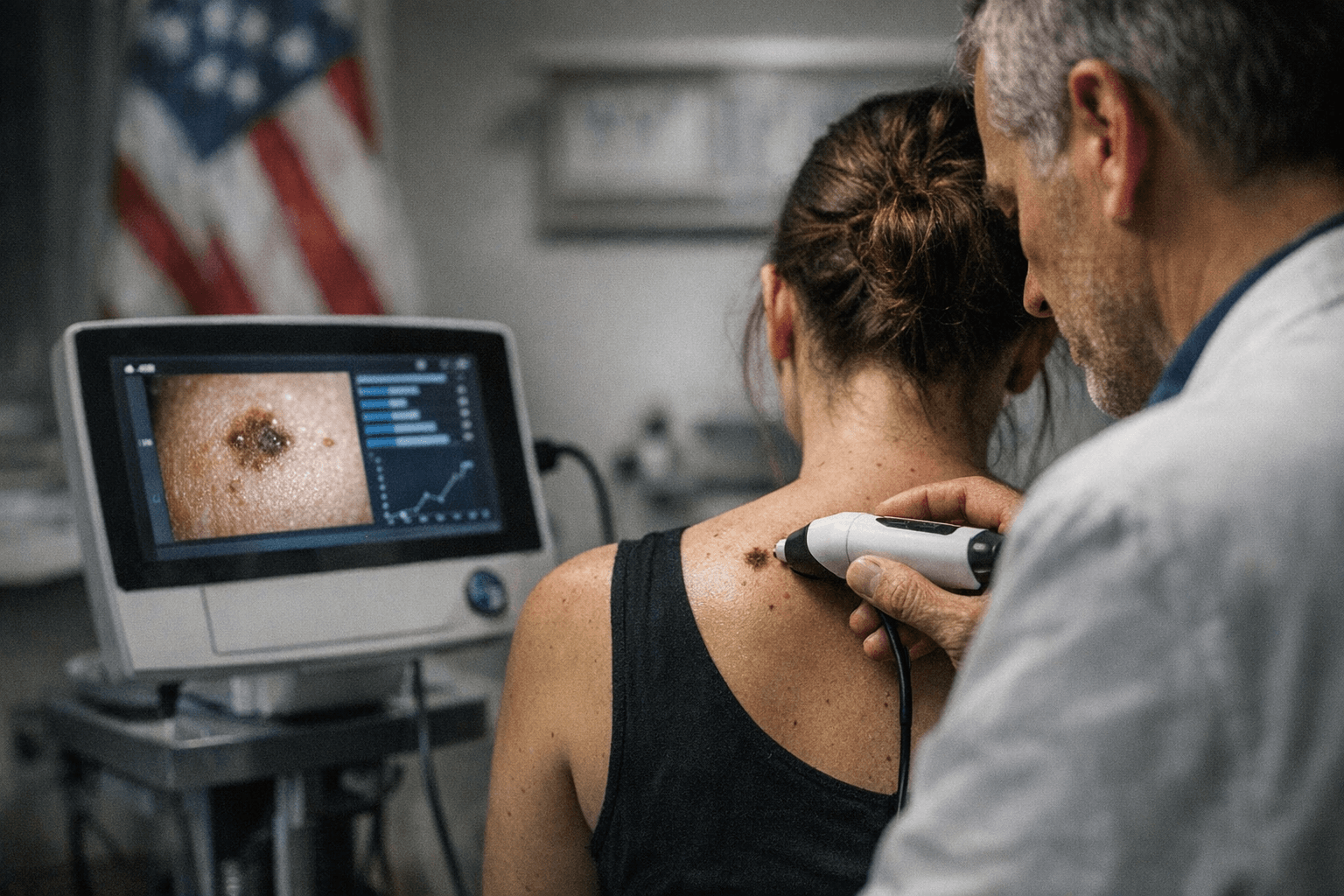
However, public health experts and health equity advocates caution that expanding device use is not a panacea. Task shifting to nonphysician clinic staff could increase access in underserved and rural areas, but it also requires robust training, quality assurance and clear protocols to avoid misdiagnosis and to ensure equitable outcomes. Deployment without parallel investment in workforce training and in affordable access risks amplifying disparities if devices concentrate in better-resourced clinics.
The company reported recent capital raises that may support commercialization. Investor materials state that a rights issue yielded approximately SEK 30.9 million and a directed issue brought in about SEK 22.5 million before issuance costs. SciBase also traces the technology’s origins to more than 20 years of research at Karolinska Institute, positioning Nevisense as a research-driven tool intended to “minimize patient suffering” and reduce healthcare costs.
The FDA action expands the regulatory pathway for a small Swedish company to push point-of-care oncology diagnostics into routine clinical settings. How quickly clinics adopt Nevisense, how payers respond, and whether broader use will translate into measurable reductions in late-stage melanoma diagnoses will depend on the specifics of the supplemental labeling, rollout plans with clinical partners, training programs and reimbursement decisions.
Know something we missed? Have a correction or additional information?
Submit a Tip

