Supira Medical Wins FDA Clearance to Launch Pivotal Heart Device Trial
FDA authorized Supira Medical's SUPPORT II trial, enrolling up to 385 patients to test a catheter-based heart pump in patients at high risk of circulatory collapse during coronary procedures.
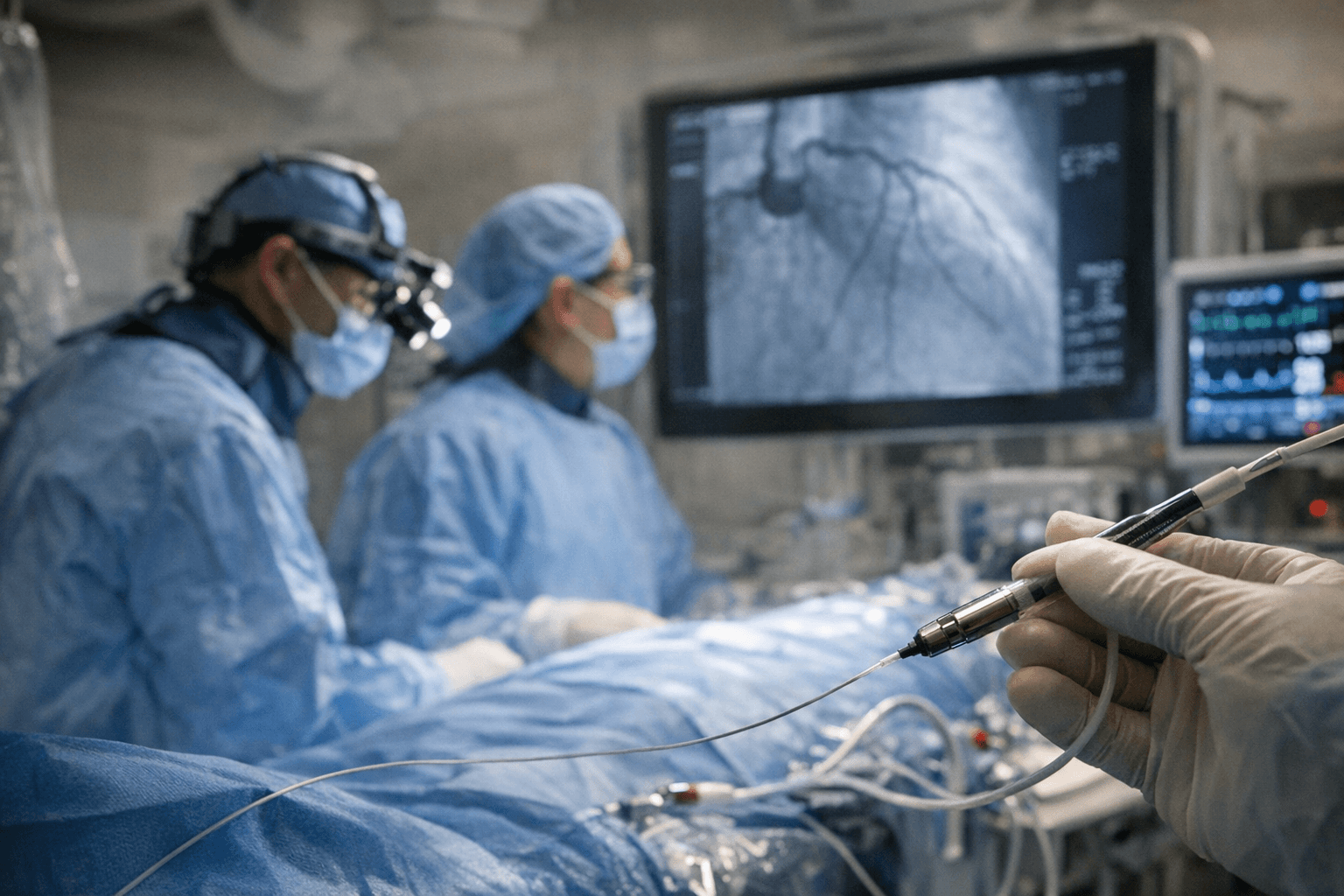
The FDA authorized Supira Medical on April 8 to begin SUPPORT II, a pivotal randomized trial of its percutaneous ventricular assist device, giving the Los Gatos, California company formal permission to generate the clinical evidence needed to seek commercial approval.
A percutaneous ventricular assist device is a catheter-delivered pump that takes over part of the heart's work when the organ cannot maintain adequate circulation. Supira's version, a 10-French profile device, is threaded through a blood vessel into the left ventricle, where it actively draws blood and delivers it to the aorta, unloading the struggling heart during high-risk coronary procedures or in cardiogenic shock, the life-threatening condition in which the heart loses the capacity to keep vital organs adequately perfused.
SUPPORT II, which expands to SUpira System in Patients Undergoing High-Risk Percutaneous COronaRy InTervention, will focus on patients who need arterial blockages opened but whose hearts are too fragile to tolerate the hemodynamic stress of the procedure without mechanical backup. The trial will enroll up to 385 patients at up to 40 U.S. sites in a prospective randomized controlled design. Dr. Ajay Kirtane of Columbia University's Vagelos College of Physicians and Surgeons and New York-Presbyterian Hospital and Dr. David Kandzari, chief of the Piedmont Heart Institute and chief scientific officer at Piedmont Healthcare in Atlanta, will serve as national co-principal investigators.
FDA authorization to initiate a pivotal trial is not product clearance. It signals that the agency accepted Supira's study design as structured to generate approvable data; a commercially available device requires a separate and more demanding step. If SUPPORT II demonstrates an acceptable safety profile alongside meaningful hemodynamic benefit, ruling out device-related harms at a rate the FDA considers tolerable, Supira can file a premarket approval application. Hospitals cannot use the device in routine clinical practice until that PMA is granted. From a coverage standpoint, payer conversations do not meaningfully begin until regulatory approval is secured.
Supira did not enter the trial without prior clinical experience. The company said its device has been used in 99 patients to date, including an international series of cardiogenic shock cases in which the device was placed via percutaneous axillary access, an approach that allowed patients to remain ambulatory during active support. That flexibility in access site, and the potential for patient mobility while on hemodynamic support, was presented as a distinguishing feature relative to existing mechanical circulatory support technologies.
The market Supira is targeting is competitive and closely scrutinized for cost-effectiveness. Established devices, including the Impella family and intra-aortic balloon pumps, already occupy catheterization labs. D. Keith Grossman, named to Supira's board in conjunction with the trial authorization, described the field as a "growing and still under-penetrated pVAD landscape," adding that he looks forward to supporting Supira as it "prepares to become a leading presence among treatment options for high-risk PCI and cardiogenic shock patients." Grossman brings 40 years of medical device experience, including specific work in mechanical circulatory support.
Dr. Nitin Salunke, Supira's president and chief executive, framed the appointment as evidence of commitment to building "an organization that is not only clinically differentiated but also positioned to execute at scale," a signal that the company views the PMA pathway as a commercial challenge as much as a regulatory one. Site initiation and patient enrollment are set to begin shortly, and how quickly Supira can fill 385 randomized slots will set the timeline for everything that follows.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip