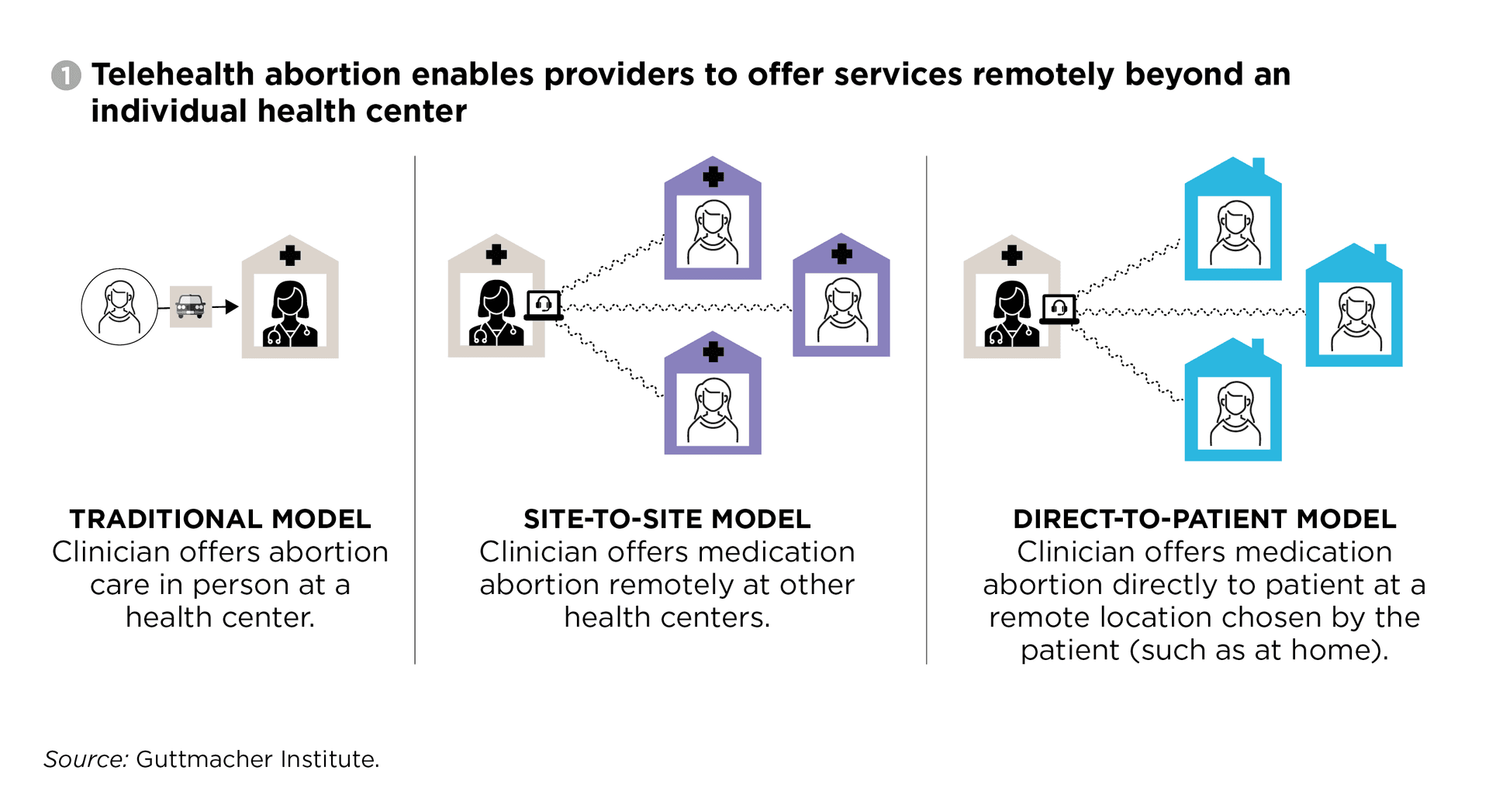
Supreme Court keeps telemedicine access to abortion pill mifepristone in place
The justices blocked a Fifth Circuit order that would have forced in-person dispensing nationwide, keeping telemedicine and mail access alive while Louisiana v. FDA continues.
The Supreme Court temporarily kept telemedicine and mail access to mifepristone in place by pausing a Fifth Circuit order that would have required in-person dispensing nationwide, even in states where abortion is legal. The case, Louisiana v. FDA, left the drug’s distribution rules in limbo while the justices weighed whether to take further action.
Mifepristone has been at the center of federal abortion policy for more than two decades. The Food and Drug Administration approved it in 2000 for use up to seven weeks of pregnancy, relaxed the rules in 2016 so it could be used through 10 weeks, prescribed by some non-physician health-care providers and dispensed after one in-person visit, and in 2021 said it would no longer enforce that initial visit requirement. The current dispute tests whether those later changes can be unwound through lower-court orders that reach far beyond Louisiana.
The Supreme Court had already confronted the drug in FDA v. Alliance for Hippocratic Medicine, deciding unanimously on June 13, 2024, that the challengers lacked standing and leaving the merits untouched. The new lawsuit reopened the fight from a different angle. Louisiana asked a federal court to impose a nationwide in-person dispensing rule that would block telehealth and pharmacy access, along with mail delivery, even where abortion remains legal. On May 1, 2026, the Fifth Circuit ruled in Louisiana’s favor. On May 4, Justice Samuel Alito entered a temporary one-week administrative stay, restoring telemedicine access while the court considered next steps.


The practical stakes are large. According to the American Civil Liberties Union, mifepristone is used in nearly two-thirds of U.S. abortions and also in early miscarriage care. SCOTUSblog and the ACLU say more than 1 in 4 abortion patients in the United States now use telemedicine. Danco Laboratories and GenBioPro, the drugmakers in the case, told the court the Fifth Circuit’s order would create immediate confusion and “regulatory chaos,” while anti-abortion groups and Louisiana argued that the FDA had unlawfully overstepped and threatened state abortion restrictions. For now, the Supreme Court has preserved the status quo, but the central question remains whether lower courts can force nationwide changes to FDA drug-distribution rules.
Know something we missed? Have a correction or additional information?
Submit a Tip