U.S. officials halt release of Covid vaccine hospital-prevention study
Federal health officials blocked a CDC study that said the latest Covid shot cut hospitalizations about 55% last winter.
Federal health officials stopped a CDC study that had been set for publication in the agency’s flagship Morbidity and Mortality Weekly Report, halting release of analysis that reportedly found the updated 2024 to 2025 Covid vaccine reduced hospitalizations by about 55% last winter. The manuscript had already been delayed after CDC acting director Jay Bhattacharya raised concerns about its methodology, turning what might have been a scientific edit into a direct intervention in the release of federal public-health research.
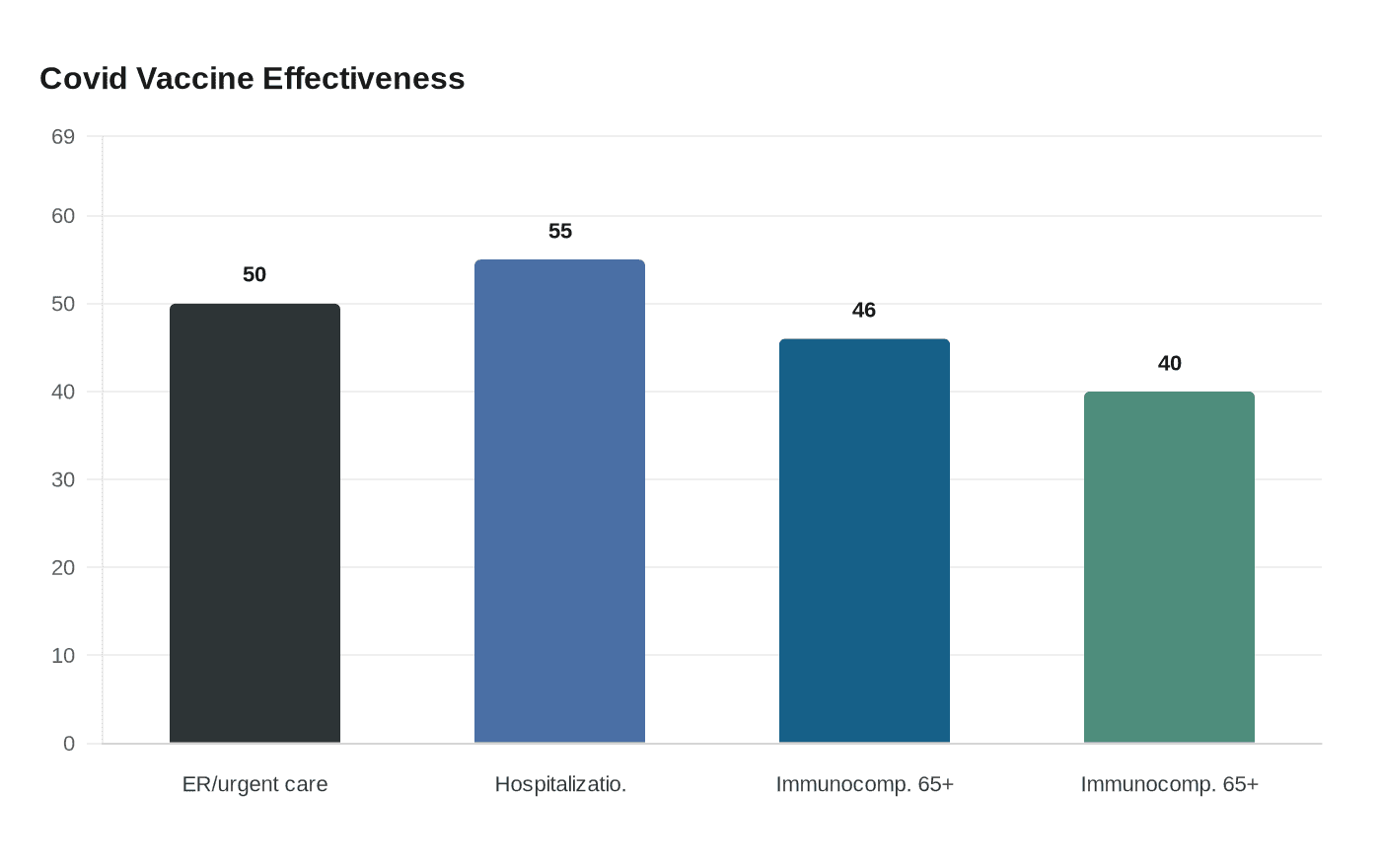
The findings at issue were not abstract. A summary of the paper said the shot reduced emergency room or urgent-care visits by about 50% and hospitalizations by about 55%, including among older adults. For clinicians caring for people at highest risk of severe illness, those numbers would have helped shape guidance for patients weighing whether vaccination still meaningfully reduced the chance of a bad outcome.

HHS spokesperson Andrew Nixon said the MMWR editorial assessment found problems with the methodological approach used to estimate vaccine effectiveness and that the manuscript was not accepted for publication. People familiar with the decision said the paper had already passed CDC scientific review before the final block, making the rejection unusual and raising questions about whether the dispute was about evidence quality or political control over how that evidence reached the public.
The study drew on the CDC’s VISION Vaccine Effectiveness Network, a collaboration established in 2019 with Westat and multiple U.S. health systems that uses linked medical, laboratory, pharmacy, billing and vaccination records. CDC says vaccine-effectiveness studies are meant to provide timely evidence to inform policy and protect public health. The agency has long used similar observational methods in flu and Covid research, including a February 2025 MMWR that reported 2024 to 2025 Covid vaccine effectiveness of 45% to 46% against hospitalization in immunocompetent adults 65 and older and 40% in immunocompromised adults 65 and older.

Bhattacharya’s role gives the episode added weight. HHS identifies him in April 2026 as both NIH director and acting CDC director, placing the decision inside a broader leadership shake-up at federal health agencies. The dispute follows other Trump administration changes to Covid policy, including the CDC ending routine Covid shot recommendations for healthy children and pregnant women in May 2025 and advisers later voting in September 2025 to limit fall vaccine eligibility to people 65 and older or those with underlying conditions.
What is at stake is larger than one manuscript. If federal officials suppress or slow publication of real-world effectiveness data, doctors can be left with less current guidance, public trust can erode further, and the credibility of agencies charged with protecting health can suffer at the very moment clear, timely evidence matters most.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip

