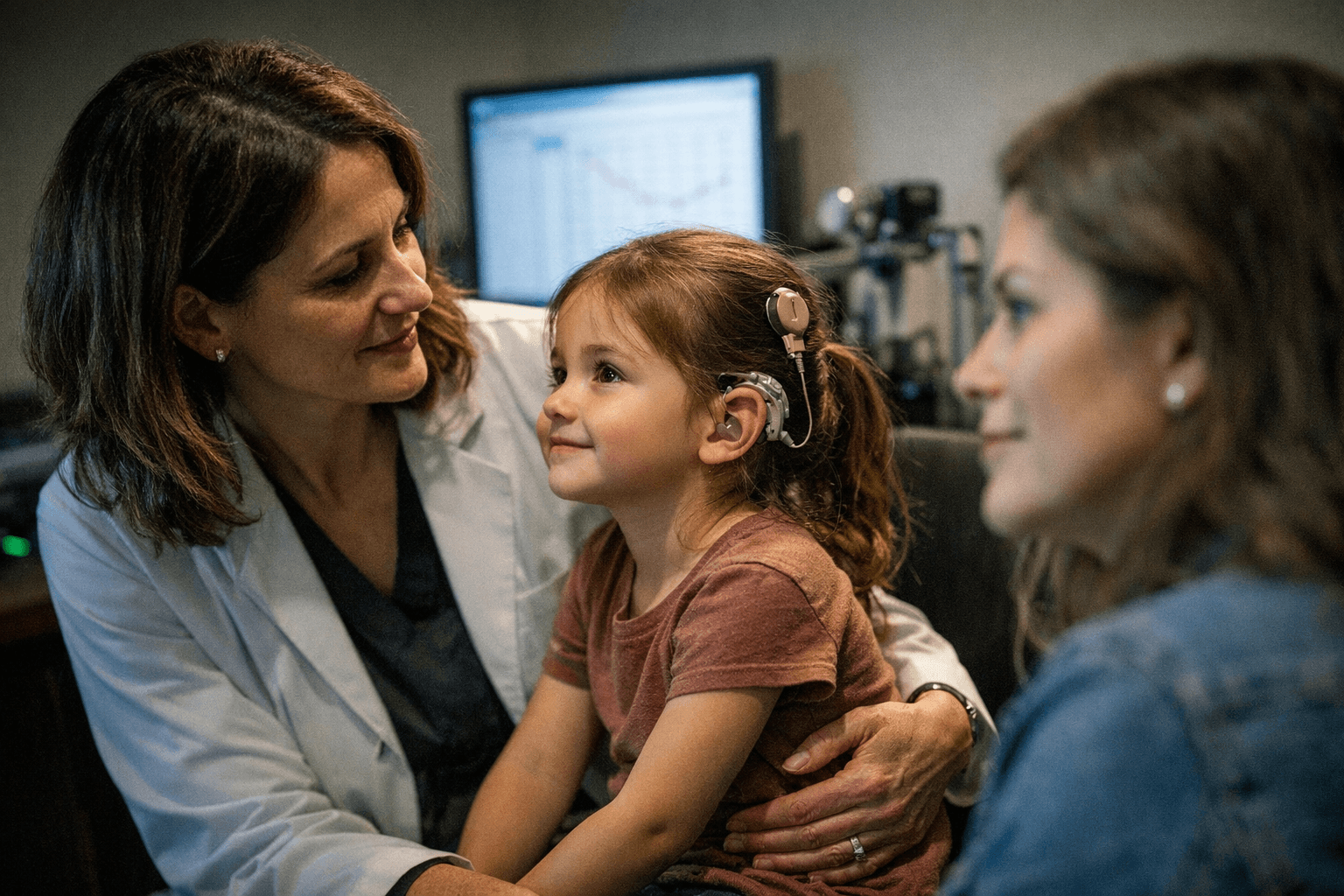FDA approves first gene therapy for inherited hearing loss
The FDA cleared the first gene therapy for inherited hearing loss, but its real test is whether children can be diagnosed early enough to benefit.

The Food and Drug Administration approved Regeneron’s Otarmeni, the first gene therapy for inherited hearing loss, opening a new front in a field that had no disease-modifying treatment for OTOF-related deafness before Thursday’s decision. The therapy, now called Otarmeni and formerly known as DB-OTO, is also the first ever dual adeno-associated virus vector-based gene therapy and the first gene therapy cleared under the Commissioner’s National Priority Voucher program.
The approval covers pediatric and adult patients with severe-to-profound or profound sensorineural hearing loss tied to biallelic variants in the OTOF gene, with preserved outer hair cell function and no prior cochlear implant in the same ear. That narrow label captures the central tension around the drug: the science is significant, but families will need a molecular diagnosis quickly enough to reach treatment before the hearing window closes. The FDA said mutations in OTOF account for 2% to 8% of inherited, non-syndromic hearing loss cases, while genetic mutations overall cause about half of congenital hearing loss. Delayed diagnosis can mean missed treatment windows and lasting speech and language delays.
The agency said the approval arrived 61 days after Regeneron filed its biologics license application, making it the sixth approval under the pilot program and tied for the fastest BLA review in modern FDA history. Otarmeni is delivered as a one-time biologic-device combination product, with a single dose per ear infused into the cochlea through a syringe and catheter connected to an infusion pump. Regeneron said it will provide the treatment for free in the United States, a rare move for a newly approved gene therapy.
The decision rested on results from the ongoing CHORD Phase 1/2 trial. Regeneron said 80% of participants met or surpassed the primary hearing endpoint at 24 weeks, and with longer follow-up 42% achieved normal hearing, including whisper-level hearing. Earlier company reporting said 12 children with profound genetic hearing loss tied to OTOF variants, ages 10 months to 16 years, had received the therapy, with nine treated in one ear and three in both ears. In that update, 10 of 11 children with at least one post-treatment assessment showed notable hearing gains.
The approval gives formal weight to a scientific idea that has advanced quickly since Regeneron began auditory gene therapy work in 2017. A 2024 report of an 11-year-old boy hearing sound for the first time helped establish that otoferlin-related deafness could be treated at the gene level, and investigators have since described responses that ranged from hearing a mother’s voice to reacting to music. For families, the bigger challenge now is practical: finding affected infants early, confirming the mutation, and getting them to a center that can deliver the therapy before time runs out.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip