FDA Approves First Gene Therapy for Genetic Hearing Loss
A first-of-its-kind gene therapy gave children with a rare OTOF mutation a chance to hear, but only a narrow group qualifies.
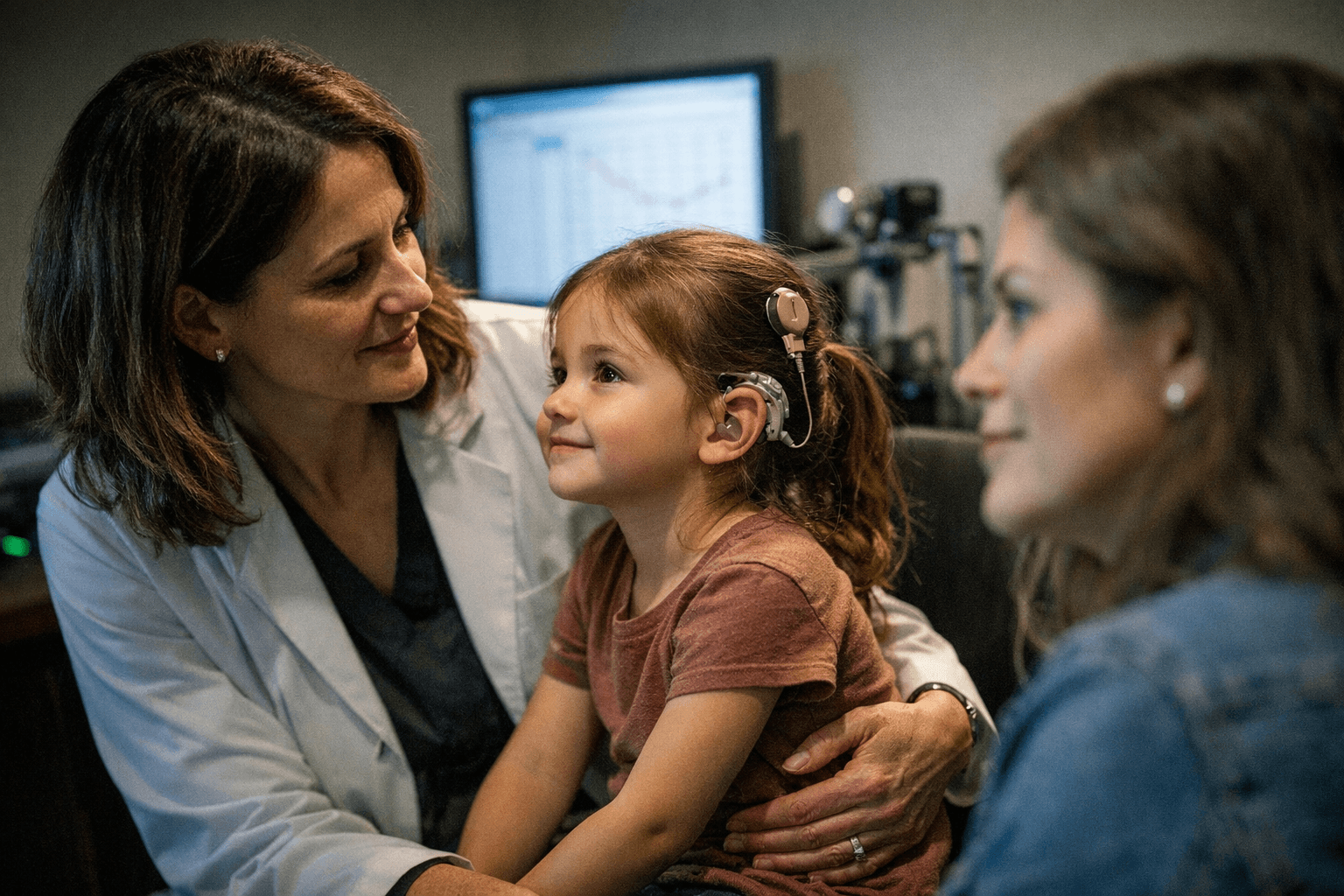
Families living with a child born deaf got an FDA milestone with sharply limited reach: the agency approved Otarmeni, the first-ever dual adeno-associated virus gene therapy, for severe-to-profound and profound sensorineural hearing loss tied to biallelic OTOF variants.
The approval, announced Thursday, April 23, 2026, covered pediatric and adult patients whose hearing loss was molecularly confirmed. The therapy targets a very specific defect. In people with two nonworking copies of OTOF, the body does not produce otoferlin, a protein needed to move sound signals from the inner ear to the brain. That makes this subtype addressable in a way many forms of hearing loss are not: the problem is not just damaged hearing, but a missing gene product that gene therapy can try to replace.
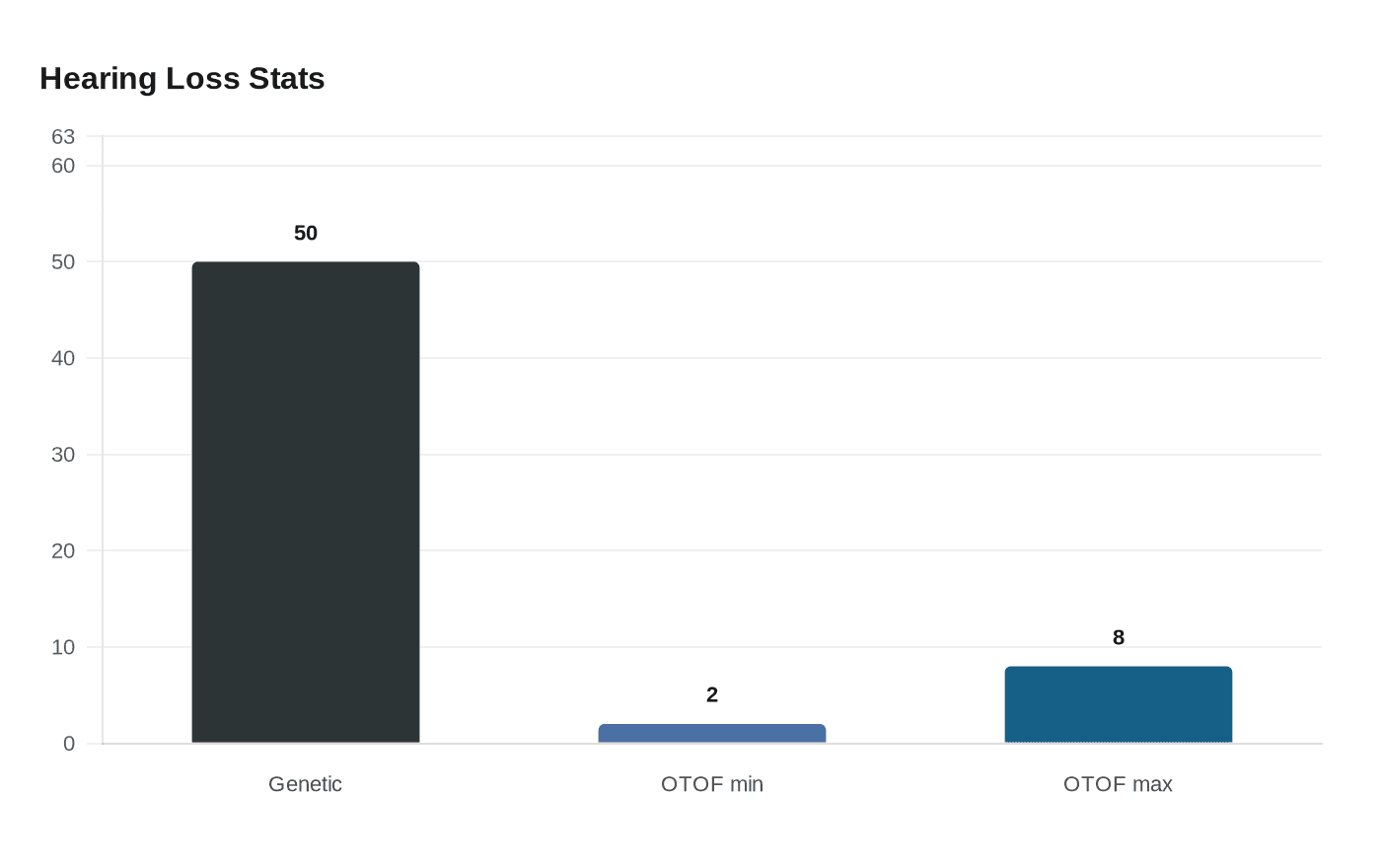
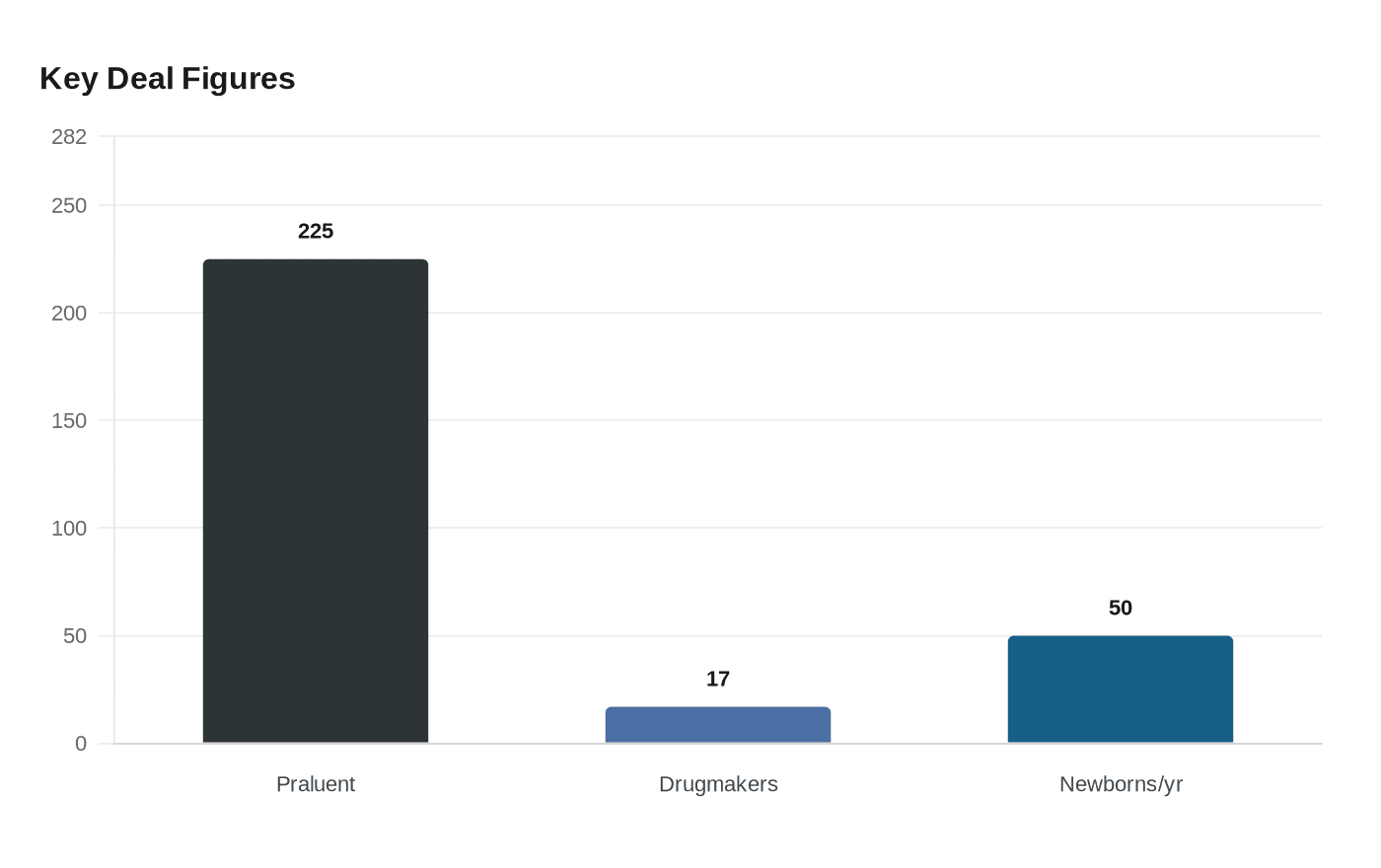
That narrow biology also explains why the drug is not a universal fix. The FDA said genetic mutations cause about half of congenital hearing loss, but OTOF variants account for only 2% to 8% of inherited, non-syndromic cases. The condition is ultra-rare, affecting an estimated 20 to 50 newborns in the United States each year. For most patients with hearing loss, the cause lies elsewhere, which means Otarmeni does not change the broader reality that hearing loss has many genetic and non-genetic forms.
The agency granted accelerated approval after hearing sensitivity improved on average pure tone audiometry at week 24. Continued approval depends on confirmatory evidence from the CHORD clinical trial. That means the therapy’s market debut carries both promise and scrutiny, with regulators still asking for proof that early gains translate into durable benefit.
Regeneron Pharmaceuticals, which acquired Decibel Therapeutics in 2023 and formerly called the treatment DB-OTO, said it will be provided free to patients in the United States. The company and clinicians have framed the approval as a turning point after years when gene therapy for hearing loss remained experimental. Earlier reports in 2024 described an 11-year-old boy who heard for the first time after OTOF-targeted therapy, a preview of the before-and-after reality now moving from study to regulated medicine.
“Our baby was born deaf, and now he can hear,” said one parent.
The FDA granted the therapy under its National Priority Voucher program, underscoring how tightly this advance is tied to both scientific precision and early genetic diagnosis. For families facing congenital deafness, timing matters as much as the treatment itself: once speech and language windows close, the lost time cannot be fully recovered.
Know something we missed? Have a correction or additional information?
Submit a Tip