Zydus Wins FDA Approval for Generic Dapagliflozin, Targets $10.2 Billion Market
Zydus Lifesciences won FDA approval for generic Farxiga in a $10.2 billion market, but uninsured patients paying $600/month won't see deep savings until the 180-day exclusivity window closes.

For patients paying out of pocket for Farxiga, brand dapagliflozin runs roughly $550 to $650 per month. The FDA's approval of the first generic versions sets a clock in motion, but when that cost actually drops at the pharmacy counter depends on competitive timing, insurer decisions, and supply readiness that no approval letter can guarantee.
Zydus Lifesciences disclosed April 8 that it received FDA final approval for generic dapagliflozin tablets in 5 mg and 10 mg strengths, a day after the agency cleared multiple generic drug applicants for the same molecule. Alembic Pharmaceuticals announced its own approval in the same period. AstraZeneca markets the branded version as Farxiga, which generated roughly $10.2 billion in U.S. annual sales through February 2026, according to IQVIA market data.
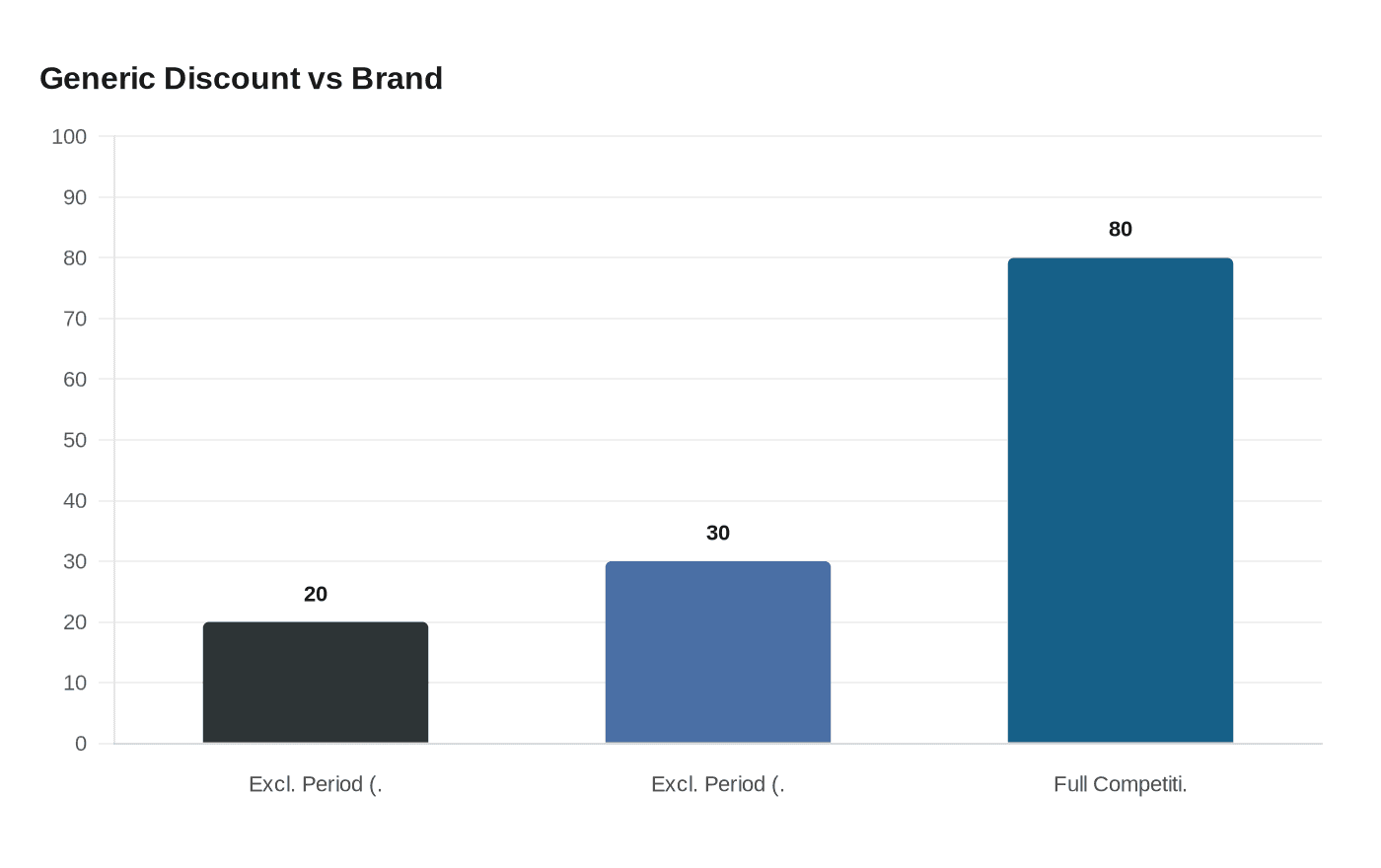
Both Zydus and Alembic qualified for 180-day shared generic exclusivity, the Hatch-Waxman mechanism that limits new competition to first-filers for six months. During that window, generics typically price 20 to 30 percent below the brand's list price rather than the steep discounts that emerge only once multi-source competition takes hold. For a drug in the $600-per-month range, that initial reduction translates to roughly $120 to $180 in monthly savings for cash-pay patients, meaningful but a fraction of what full generic competition eventually delivers.

The sharper savings require the 180-day exclusivity clock to run its course. On comparable high-volume branded drugs, generic prices have historically fallen 80 percent or more below brand once multiple manufacturers enter the market. That phase also demands sufficient production capacity across suppliers, and Zydus will manufacture the tablets at its formulation facility inside the Ahmedabad Special Economic Zone, a site already integrated into the U.S. generics supply chain.
Even after commercial launch, most insured patients won't see the benefit immediately. Pharmacy benefit managers update formulary tiers on quarterly or annual cycles, meaning generic dapagliflozin may not appear as a preferred lower-cost alternative for three to six months after it reaches wholesalers. Until a PBM formally substitutes the generic on formulary, patients with commercial insurance will largely continue filling Farxiga at brand-tier cost-sharing levels.
The clinical breadth of dapagliflozin adds another variable to the coverage timeline. Beyond glycemic control in type 2 diabetes, it carries FDA-approved indications for reducing hospitalization risk in adults with heart failure and established cardiovascular disease, and it is used in chronic kidney disease management. Whether insurers extend generic formulary preference uniformly across all three indications, or tier them separately, will determine which patient populations benefit earliest and most directly.
For Zydus, the approval deepens a U.S. generics portfolio positioned to capture share in a market large enough to generate substantial revenue even at significantly reduced per-unit pricing. For AstraZeneca, the transition initiates the familiar branded pharmaceutical response: rebate restructuring, outcomes-based contracting, and potential litigation strategies aimed at sustaining Farxiga's commercial position as generic entrants press for formulary placement.
The 180-day exclusivity window will serve as the first real test of how quickly Zydus and Alembic can convert FDA clearance into commercial momentum, and whether additional generic applicants emerge before the broader market fully opens.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip

