Abbott Loses $70 Million Verdict Over Premature Infant Formula NEC Claims
A unanimous Cook County jury awarded $70M against Abbott over Similac Special Care NEC claims, with $17M of that in punitive damages targeting the company's conduct directly.

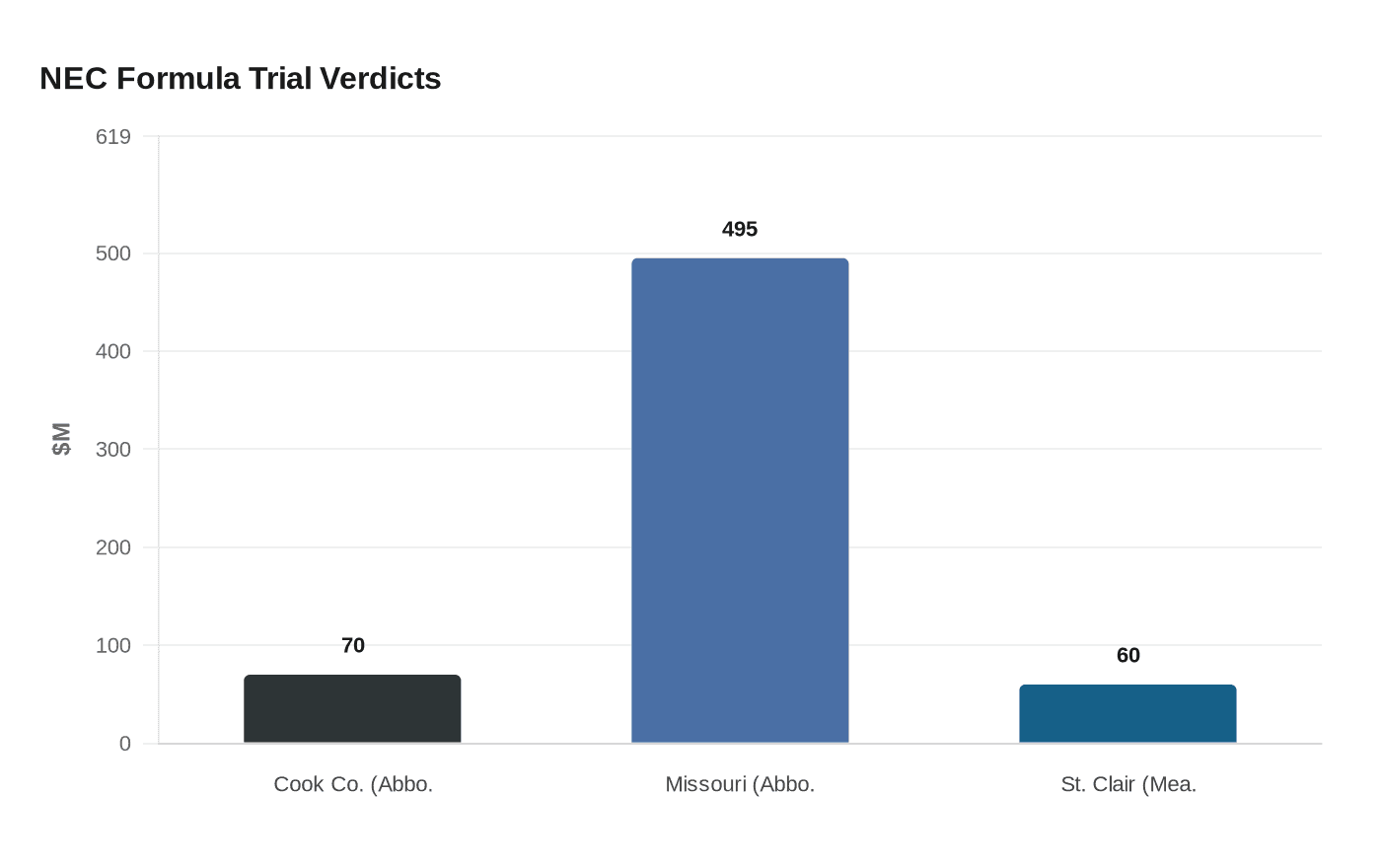
A unanimous Cook County jury found Abbott Laboratories liable for injuries suffered by four premature infants fed its Similac Special Care formula in hospital intensive care units, awarding their families $70 million. The verdict arrived across two days: $53 million in compensatory damages on Thursday, followed by $17 million in punitive damages on Friday, April 10.
The case centered on necrotizing enterocolitis, a severe and sometimes fatal gastrointestinal disease that primarily strikes preterm infants. Attorneys from the firm Keller Postman argued that Abbott was aware cow's milk-based formula raises NEC risk in neonatal intensive care settings but failed to warn the clinicians and families whose newborns received it. Jurors sided with the families on claims of defective design, failure to warn, and negligence. All four infants survived, though each suffered serious complications and three required surgery shortly after birth.
"The jury's verdicts on behalf of these four infants confirm once again what Abbott has known for years and chosen to ignore," said Ben Whiting, Senior Partner at Keller Postman. Keller Postman partner Amelia Frenkel added: "For decades, Abbott Laboratories has shown a flagrant disregard for the health of the most fragile infants. Safety has to come first for every preterm baby born today and tomorrow and into the future."

Abbott disputed the verdict and said it plans to appeal, maintaining that medical experts and regulators consider Similac Special Care safe and necessary for premature infants. The company has warned that mounting jury awards could restrict treatment options in neonatal care by increasing liability exposure for manufacturers.
The Cook County result is the third major trial loss in NEC formula litigation and Abbott's second. In 2024, a Missouri jury in the 22nd Judicial Circuit Court in St. Louis returned a $495 million verdict against Abbott; a separate Illinois jury in St. Clair County hit Mead Johnson, maker of Enfamil, with a $60 million verdict. Thousands of additional lawsuits against both manufacturers remain active. The legal pressure intersects with ongoing FDA and CDC scrutiny of formula labeling, contamination controls, and supplier surveillance practices. Policy analysts have argued that verdicts of this magnitude sharpen regulatory momentum toward stricter labeling mandates and ingredient testing requirements across the supply chain.
For parents adding formula to registries or navigating gift conversations at showers, context matters here. The verdict specifically targets Similac Special Care, a product formulated for premature infants in clinical settings, not standard over-the-counter formula for full-term babies. If you have a preterm infant or anticipate one, ask your neonatologist directly which feeding products are in use, what NEC risk disclosures apply, and whether donor human milk is available in your NICU. Before adding any formula to a registry, verify current lot safety through the FDA's recall database and choose retailers that offer lot-number tracking and clear exchange policies if a recall follows purchase. Cash-contribution and diaper-fund registries sidestep product liability uncertainty entirely and give new parents the flexibility to buy through channels they trust. When the topic surfaces at a shower, the framing is straightforward: formula is a medical decision that belongs between parents and their care team, not a referendum on parenting choices.
With thousands of NEC claims still pending and Abbott's appeal ahead, the pressure on manufacturers to overhaul labeling and clinical communication is not going to ease.
Sources:
Know something we missed? Have a correction or additional information?
Submit a Tip